Fingerprinting uranium: X-rays identify mobile, stationary forms of atomic pollutant

(PhysOrg.com) -- Determining if uranium will zip through the soil or not is easier now, thanks to scientists at Pacific Northwest National Laboratory and the University of North Texas. Dr. Eugene Ilton and Dr. Paul Bagus elucidated a systematic approach for identifying uranium's state and hence its mobility. Their method uses x-rays, in the form of x-ray photoelectron spectroscopy. Their technique and detailed analysis appear in an invited perspective article in Surface and Interface Analysis.
"This is the first time that anyone has formalized this approach and showed how broadly useful it is," said Dr. Eugene Ilton, a PNNL geochemist.
Uranium poses risks around the world. At one U.S. site, 200,000 kg or about 220 tons of uranium entered the environment. Whether the radionuclide travels to nearby water sources depends on its oxidation state, the number of electrons around the atom. Ilton and Bagus' method accurately pinpoints uranium's oxidation state. Using this method allows scientists to more accurately predict the atomic pollutant's behavior.
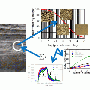
This technique begins with XPS, which is used commercially and scientifically to determine the chemical state, elemental composition, and other details about materials. The instrument directs a beam of X-rays at the sample, which is placed under ultra high vacuum. The beam excites electrons within the sample, forcing the electrons to leap off. Sensors within the instrument measure the number of electrons that escape at a particular kinetic energy.
The data is plotted as the number of electrons versus kinetic energy, or binding energy, forming peaks that are characteristic of an element in the material. The kinetic energy and binding energy are related by a simple formula discovered by Albert Einstein. Scientists can interpret the size, shape, and energy of the peaks to characterize the samples. The peaks fall roughly into two categories: primary and secondary. The primary peaks are usually larger than the secondary peaks, which are called satellites.
Ilton and Bagus analyzed samples containing very dilute amounts of uranium using the XPS in EMSL and also did a broad literature search on previous XPS work on uranium compounds. They focused on the satellite structures and showed that the energy separation between the satellite and primary peaks was diagnostic of uranium oxidation states over a broad range of compounds with very different compositions.
This led to the development of a methodical process for taking XPS data and determining the oxidation state of uranium. While the team was not the first to use uranium satellite structures in this way, they showed that the method is broadly applicable to a wide range of uranium-bearing materials.
"Using XPS can provide a definitive fingerprint of the oxidation state of uranium," said Ilton.
Ilton and Bagus are working on theoretical approaches to pull more information from the XPS spectra. The data they provide can be used in other studies examining the oxidation state of uranium and other metal compounds.
More information: ES Ilton and PS Bagus. 2011. "XPS Determination of Uranium Oxidation States." Surface and Interface Analysis 143(13):1549-1560. DOI: 10.1002/sia.3836
Provided by Pacific Northwest National Laboratory