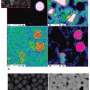
Uranium adsorption in sediments varies with respect to grain size
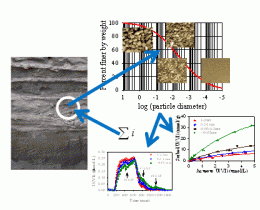
Using experimental and modeling resources at EMSL, scientists from the Pacific Northwest National Laboratory determined the equilibrium and kinetic properties of uranium(VI) adsorption to subsurface grains, which ranged in size from tiny clay particles to larger pieces of gravel. They found that the kinetics or the rate of adsorption onto the grains varies with respect to the grain size. Specifically, the larger-size grains adsorbed less uranium and did so at a slower rate than smaller grains. After determining the size-specific equilibrium and kinetic properties, the team created a series of mathematical formulas that allowed them to calculate uranium adsorption in a heterogeneous subsurface sample from the Hanford Site, a plutonium production complex in southeastern Washington State.
Uranium and other contaminants exist across large swaths of the Hanford Site subsurface. Calculating exactly how much uranium is present requires scientists to determine the adsorption behavior of the subsurface grains in the laboratory and then scale that knowledge to the field. This process has proven difficult for researchers in the past because of the heterogeneity of grain size distribution at the field.
The scientists on this study began by conducting a series of wet chemistry experiments to determine adsorption characteristics of different grain sizes. The experimental resources included surface area and microporosity instruments as well as stirred flow cells and other instrumentation. Following on the laboratory results, computer simulations were done to quantify the kinetics and equilibrium thermodynamics. Then, the team developed appropriate formulas for adding the adsorptions to predict the uranium held in a larger sample.
The team tested their approach using a sample of contaminated Hanford Site sediment. Their approach agreed well with the measurements. The team is continuing to study this approach and will be moving on to determine the applicability to groundwater-infused sediments.
More information: Shang, J, et al. 2011. “Effect of Grain Size on Uranium(VI) Surface Complexation Kinetics and Adsorption Additivity.” Environmental Science & Technology 45(14):6025-6031. DOI: 10.1021/es200920k
Provided by Environmental Molecular Sciences Laboratory