Carbon dioxide recycling? 'Diagonal' approach for reductive functionalization of carbon dioxide
(PhysOrg.com) -- Carbon dioxide is a by-product of energy production, but must it always be viewed as a waste product? This gas could be a useful renewable resource and an environmentally friendly chemical reagent. If we really could use it, it would not just reduce the emission of carbon dioxide into the atmosphere, but also our dependence on petrochemicals, which will eventually start to run out. In the journal Angewandte Chemie French scientists working with Thibault Cantat at the Institut Rayonnement Matière de Saclay in Gif-sur-Yvette have now introduced a new approach for the conversion of carbon dioxide into both useable building blocks for chemical synthesis and new fuels.
“Carbon dioxide is a nontoxic, abundant C1 building block,” says Cantat. “Only a handful of processes using this starting material have been developed, because carbon dioxide is a very stable molecule that can not easily be made to react.” To date, there have been two different approaches for the use of carbon dioxide. According to Cantat, “In the ‘vertical’ approach, the carbon dioxide is reduced, which means that the oxidation state of the carbon atom is reduced by the formal replacement of oxygen with hydrogen. This results in molecules such as methanol or formic acid, which can be converted into fuels.” These products have a higher energy content than carbon dioxide, but only a handful of chemicals can be produced this way.
“In the ‘horizontal’ approach, the carbon atom is functionalized, which means that it forms new bonds to oxygen, nitrogen, or other carbon atoms”, continues Cantat. “The oxidation state stays the same, the energy content is not increased.” This does not produce fuels, but chemicals that are useful building blocks for chemical syntheses, such as urea.
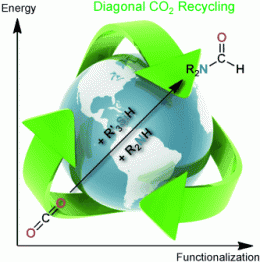
The French team thus tried a compromise approach, a combination of both methods to make a “diagonal” approach. By their method, the carbon dioxide is both reduced and functionalized in one step. This allows the synthesis of a much greater number of chemicals, directly from CO2.
This reaction requires three things: a reducing agent (e.g. a silane), an organic molecule to be attached to the carbon atom of the carbon dioxide (e.g. an amine), and a special catalyst that catalyzes both the reduction and the functionalization. The successful catalyst is a special organic base consisting of a nitrogen-containing ring system. “Variation of the reaction partners should allow us to make a whole series of chemical compounds that are normally obtained from petrochemical feedstocks,” says Cantat, “for example, formamide derivatives, which are important intermediates for both chemical and pharmaceutical industries.”
More information: Thibault Cantat, A Diagonal Approach to Chemical Recycling of Carbon Dioxide: Organocatalytic Transformation for the Reductive Functionalization of CO2, Angewandte Chemie International Edition, dx.doi.org/10.1002/anie.201105516
Provided by Wiley