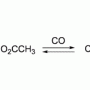Researchers stumble on colorful discovery

Modified metals that change colour in the presence of particular gases could warn consumers if packaged food has been exposed to air or if there's a carbon monoxide leak at home. This finding could potentially influence the production of both industrial and commercial air quality sensors.
"We initially found out by accident that modified rhodium reacts in a colourful way to different gases," says Cathleen Crudden, a professor in the Department of Chemistry. "That happy accident has become a driving force in our work with rhodium."
Rhodium that is modified using carbon, nitrogen or hydrogen–based complexes changes to yellow in the presence of nitrogen, deep blue in the presence of oxygen, and brown in the presence of carbon monoxide.
This colour change occurs because of the way that the gases bind to the compound's central metal, according to the researchers.
Another remarkable aspect of this discovery is that the chemical changes take place without disrupting the exact placement of each individual atom in the compound's crystalline lattice. Dr. Crudden notes that this type of transformation is virtually unprecedented.
Rhodium is the main metal used in the production of catalytic convertors to reduce the toxicity of car exhaust emissions. Dr. Crudden's team, including graduate student Eric Keske and postdoctoral fellow Dr. Olena Zenkina, is currently investigating whether cobalt, a significantly cheaper metal than rhodium, reacts similarly.
Provided by Queen's University