From the rustbelt: An iron-based flow battery
Researchers at Case Western Reserve University are mixing cheap and plentiful iron in benign solutions to create a flow battery – essentially an unwrapped battery that can be scaled up to hold and supply electricity to a home or an entire community.
The goal is to produce a cheap and efficient system capable of storing energy from wind turbines and solar panels and supplying energy when wind wanes and the sun sets. The battery could also be integrated into a smart grid, charging up when usage is low then adding electricity when need is high.
Not only will the flow battery be cheaper and more efficient than current models, but much more environmentally friendly, the researchers say.
"We like to call this the rustbelt battery," said Robert Savinell, professor of chemical engineering at Case Western Reserve.
Savinell proposed an iron-based flow battery 30 years ago but the energy industry was more interested in other technologies. Now, with the move toward sources of intermittent energy and increased gird efficiency, such a storage system could be one answer to 21st-century needs.
Savinell and Jesse Wainright, a fellow chemical engineering professor at Case School of Engineering, have begun a three-year project to fine tune the chemistry, develop the cleanest, most efficient system, and build a working model proving the technology.
The Department of Energy's Office of Electricity Delivery and Energy Reliability, through Sandia National Laboratory, is funding the research with a $600,000 grant.
For large-scale energy storage, a flow battery has significant advantages over a standard battery.
In standard batteries, power and energy densities are limited by wrapping all the materials used to convert chemical energy into electrical energy inside of a single cell. The electrodes, which are part of the fuel, are consumed over time, leading to performance loss.
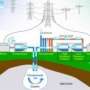
In flow batteries, chemical reactants used to produce electrical energy are stored in two tanks and the electrodes, which are not used as fuel, are housed in a separate chamber. The reactants are pumped one direction through the chamber to charge the battery and the other direction to discharge the system.
Power and energy density can be increased by increasing the volume of reactants.
The most common flow batteries are based on vanadium, a metal mined primarily in Russia, China and South Africa, and which has recently cost from $8 to $20 per pound in the pentaoxide form. Iron, which is plentiful in the U.S., has recently been selling for less than 25 cents per pound as anhydrous ferrous chloride, or on a metal basis less than 1% of the cost of vanadium.
Vanadium batteries use highly-corrosive sulfuric acid for the electrolyte. For safety reasons, the researchers plan to use a benign electrolyte with a pH of about 4.
"Since these systems will be very large, we're very conscious of the hazards that could arise from an accident," Wainright said. "We're focusing our efforts on developing a safe chemistry; I wouldn't want to put anything in the battery that you couldn't swim in."
A large-scale energy storage facility that could accommodate a wind farm by storing up to 20 megawatt-hours of electricity would require two storage tanks for the iron solutions of about 250,000 gallons – or 8 railroad tank cars each, he explained.
A system that size could supply the power needs of 650 homes for a day.
Flow batteries can be a useful alternative to storage technologies such as pumped hydro and compressed air systems, which require large water supplies and land with mixed elevations, or access to airtight caverns, Savinell said.
When demand is low, pumped hydro stations use excess electricity to pump water from a river or reservoir to a reservoir at a higher elevation. When demand rises, the water is released downhill through turbines that produce electricity. Compressed air stations pump air into caverns when demand is low then release the compressed air through turbines to produce electricity as demand increases.
The efficiency of the systems can reach about 75 percent. Savinell and Wainright estimate the iron flow battery can reach 80 percent.
Sandia set a goal of creating new kinds of storage systems that would cost $100 per kilowatt-hour produced.
The researchers estimate, because of the low cost of components, that the iron-based battery would cost $30 per kilowatt-hour.
Provided by Case Western Reserve University