March 6, 2009 weblog
Liquid Battery Offers Promising Solar Energy Storage Technique

(PhysOrg.com) -- One of the biggest challenges currently facing large-scale solar energy technology is finding an effective way to store the energy, which is essential for using the electricity at night or on cloudy days.
Recently, researchers from MIT have designed a new kind of battery that, unlike conventional batteries, is made of all-liquid active materials. Donald Sadoway, a materials chemistry professor at MIT, and his team have fabricated prototypes of the liquid battery, and have demonstrated that the materials can quickly absorb large amounts of electricity, as required for solar energy storage.
"No one had been able to get their arms around the problem of energy storage on a massive scale for the power grid," says Sadoway. "We're literally looking at a battery capable of storing the grid."
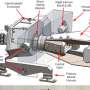
The battery consists of three layers of liquids: two electrode liquids on the top and bottom (electrodes are usually solid in conventional batteries), and an electrolyte liquid in the middle. In the researchers' first prototype, the electrodes were molten metals - magnesium on the top and antimony on the bottom - while the electrolyte was a molten salt such as sodium sulfide. In later prototypes, the researchers investigated using other materials for improved performance.
Since each liquid has a different density, the liquids automatically form the three distinct layers. When charging, the solid container holding the liquids collects electrons from exterior solar panels or another power supply, and later, for discharging, the container carries the electrons away to the electrical grid to be used as electricity.
As electrons flow into the battery cell, magnesium ions in the electrolyte gain electrons and form magnesium metal, rising to form the upper molten magnesium electrode. At the same time, antimony ions in the electrolyte lose electrons, and sink to form the lower molten antimony electrode. At this point, the battery is fully charged, since the battery has thick electrode layers and a small layer of electrolyte. To discharge the electrical current, the process is reversed, and the metal atoms become ions again.
As Sadoway explained in a recent article in MIT's Technology Review, the liquid battery is a promising candidate for solar energy storage for several reasons. For one thing, it costs less than a third of the cost of today's batteries, since the materials are inexpensive and the design allows for simple manufacturing. Further, the liquid battery has a longer lifetime than conventional batteries, since there are no solid active materials to degrade. The liquid battery is also useful in a wide range of locations compared with other proposed solar storage methods, such as pumping water. Most importantly, the liquid battery's electrodes can operate at electrical currents tens of times higher than any previous battery, making it capable of quickly absorbing large amounts of electricity.
The researchers hope to commercialize the liquid battery in the next five years. As Sadoway explained, connecting the batteries into a giant battery pack to supply electricity for New York City would require nearly 60,000 square meters of land. Such a battery pack could store energy from enormous solar farms, which would replace today's power plants and transmission lines as they become old.
via: Technology Review
© 2009 PhysOrg.com