Green and lean: Secreting bacteria eliminate cost barriers for renewable biofuel production

A Biodesign Institute at Arizona State University research team has developed a process that removes a key obstacle to producing low-cost, renewable biofuels from bacteria. The team has reprogrammed photosynthetic microbes to secrete high-energy fats, making byproduct recovery and conversion to biofuels easier and potentially more commercially viable.
"The real costs involved in any biofuel production are harvesting the goodies and turning them into fuel," said Roy Curtiss, of the Institute's Center for Infectious Diseases and Vaccinology and professor in the School of Life Sciences. "This whole system that we have developed is a means to a green recovery of materials not requiring energy dependent physical or chemical processes."
Curtiss is part of a large, multidisciplinary ASU team that has been focusing on optimizing photosynthetic microbes, called cyanobacteria, as a renewable source of biofuels. These microbes are easy to genetically manipulate and have a potentially higher yield than any plant crops currently being used for the production of transportation fuels.
But, until now, harvesting the fats from the microbes has required many costly additional processing steps that contribute up to 70 to 80 percent of the total cost of their renewable biofuel production, making them uncompetitive compared with petroleum production costs.
Cyanobacteria have a tough, protective set of outer membranes that help the bacteria thrive in even harsh surroundings, creating the pond scum often found in backyard swimming pools. Like plants, they are dependent upon sunlight, water and carbon dioxide for growth.
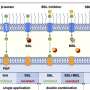
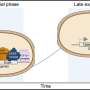
To get cyanobacteria to more readily release their precious, high fat cargo, Curtiss and postdoctoral researcher Xinyao Liu, placed a suite of genes into photosynthetic bacteria that produced enzymes to degrade membrane lipids, poking holes in the membranes to release free fatty acids into the water. In a clever feat of genetic reprogramming of the cells, the enzymes are only produced when carbon dioxide ---a vital ingredient of bacterial growth---is removed from their environment.
"We first freed up fatty acids by triggering self-destruction of the bacteria by adding nickel," Liu said, "but this is not so good for the environment. So, this time we did it in a smarter way – by stopping carbon dioxide supply. The strategy of adding nothing for recovering fuels from biomass is designed to drastically reduce processing costs."
"Genetics is a very powerful tool," added Liu, who recently presented the results at the 3rd Annual World Algae Summit in San Diego, California. "We have created a very flexible system that we can finely control. After teaching cyanobacteria to excrete fuels, we don't want to waste the useful lipids in the photosynthetic membranes, so we developed a greener way to recycle the remaining value of the biofactory."
The team tested fat-degrading enzymes, called lipases, from bacterial, fungal and guinea pig sources to see which would work best. These lipases are able work like molecular scissors, clipping off the fatty acids from the photosynthetic membranes. They also worked to optimize the growth conditions of their green recovery method, testing variables such as the cell culture density of the microbes, light intensity and agitation of the cultures.
The team's ingenuity rests in part with their ability to utilize the full repertoire of nature's toolkit. "Due to rapid DNA sequencing and public gene databases, we can now use this vast and ever-increasing store of gene sequences with powerful computer search methods to identify the best genes and proteins with optimal functions and capabilities independent of their origin in microbes, plants and animals," said Curtiss. "It is like being a kid in a candy store the size of the State of Arizona and finding the most delicious candy treat almost in the time to snap your fingers!"
The project is also a prime example of the multidisciplinary, collaborative spirit of ASU research combining the expertise of bacteriologists, molecular biologists and engineers. Other key contributors were Biodesign colleagues Sarah Fallon and Jie Sheng.
Next, the group will test their results in large-scale photobioreactors, which are being designed by engineers in the institute's Swette Center for Environmental Biotechnology to optimally capture the free fatty acids. Ultimately, the team hopes to achieve development of a new, economical and environmentally friendly, carbon neutral source of biofuels.
"We are optimistic that we can make the system even better, leading to the commercialization of our green recovery method bundled with other technologies," said Liu.
The project has been part of the state of Arizona's strategic research investments to spur new innovation that may help foster future green and local industries. The state's abundant year-round sunshine and warm temperatures are ideally suited for growing cyanobacteria.
More information:
The results were published in the early online edition of the Proceedings of the National Academy of Sciences www.pnas.org/cgi/doi/10.1073/pnas.1103016108
Print version: Proc. Natl. Acad. Sci. USA. 108:6905-6908.
Provided by Arizona State University