Novel approach uses ion-molecule collisions, deposition to create sought-after material

(PhysOrg.com) -- Once only possible with expensive liquids and large amounts of waste, scientists can now create an efficient, easy-to-separate catalyst with small amounts of material, thanks to an innovative approach from Pacific Northwest National Laboratory. Dr. Julia Laskin and Dr. Grant Johnson employed collisions between ions and molecules within a mass spectrometer, a popular analytical technique, to manipulate ruthenium-centered ions in the gas phase. The resulting highly reactive ions, which cannot be generated easily in solution, were then gently deposited onto a selected surface. The product is a catalytically active material that may one day be used in fuel cell and solar energy storage applications.
"Using the unique capabilities of a mass spectrometer we can manipulate an ion in the gas phase and obtain a material that is unattainable in solution," said Johnson, a physical chemist and a Linus Pauling Distinguished Postdoctoral Fellow at PNNL.
When it comes to producing fuels, whether bio- or petroleum-based, catalysts drive the reactions, thereby reducing cost, inefficiency, and waste. Scientists at PNNL and elsewhere are working to design catalysts that combine the selectivity of homogenous catalysts with the ease of use of heterogeneous catalysts. Heterogeneous catalysts, which are less selective, are commonly used because they are easy to separate from the reactants and products. This study shows a new approach that allows scientists, for the first time, to create unique, easy-to-separate catalysts.
Laskin and Johnson selected the ruthenium tris(bipyridine) complex to study because of its applications in solar energy conversion and oxidation catalysis. The complex has a ruthenium metal atom surrounded by a set of three ligands, strings of atoms that wrap around the ruthenium center. With all three wrappers in place, the catalyst is fairly unreactive. However, removing one of the wrappers and partially exposing the metal center makes the complex catalyze reactions that turn simple chemical feedstock, such as ethylene, into valuable chemical intermediates such as ethylene oxide. "This ruthenium complex has been a hot topic lately," said Laskin.
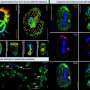
In this study, the scientists began by putting the ruthenium-centered catalyst into the gas phase using electrospray ionization. Then, the ions were focused through an ion funnel. They accelerated the ion beam to cause the ions to collide with gas molecules inside the vacuum chamber. The collisions caused the ligands to snap off, exposing the catalytically active ruthenium core. The ions were then passed through powerful oscillating electric fields that separated out the desired unwrapped ruthenium ions.
The researchers directed a beam of the desired ruthenium ions at a surface, a carboxylic acid terminated self-assembled monolayer atop a very thin layer of gold. They transferred the surface into a time-of-flight secondary ion mass spectrometer (TOF-SIMS). Using the data from the in situ TOF-SIMS, they determined that the unwrapped ruthenium-centered catalysts adhere strongly to the surface and exhibit behavior that is consistent with the catalytic oxidation of ethylene to ethylene oxide.
The team is continuing to investigate the possibilities of generating new catalytic species in the gas phase and depositing them onto surfaces. This work will further the controlled preparation of materials that are necessary for advancements in catalysis and solar energy conversion.
More information: Johnson GE and J Laskin. 2010. "Preparation of Surface Organometallic Catalysts by Gas-Phase Ligand Stripping and Reactive Landing of Mass-Selected Ions." Chemistry: A European Journal 16, 14433-14438.
Provided by Pacific Northwest National Laboratory