Fast, easy way to make hydrogen nanosensors found by scientists
A team of Northern Illinois University scientists, with a major role played by NIU Ph.D. students, has discovered a new, convenient and inexpensive way to make high performance hydrogen sensors using palladium nanowires.
The technology could help enable a scale-up for potential industrial applications, such as safety monitors in future hydrogen-powered vehicles.
Highly flammable hydrogen gas cannot be odorized like natural gas. The new technology produces nanoscale sensors that work extremely fast and would allow for closing of safety valves before dangerous concentrations of the gas could be reached.
Scientists have known that palladium nanowires demonstrated promise as hydrogen gas sensors in speed, sensitivity and ultra-low power consumption. But the utilization of single palladium nanowires faced challenges in several areas, including nanofabrication.
“We report on hydrogen sensors that take advantage of single palladium nanowires in high speed and sensitivity and that can be easily and cheaply made,” said lead author Xiaoqiao (Vivian) Zeng, a Ph.D. student in chemistry and biochemistry at NIU. The new research is published in the January edition of the American Chemical Society's prestigious journal Nano Letters.
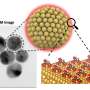
“The new types of hydrogen sensors are based on networks of ultra-small (< 10 nanometers) palladium nanowires achieved by sputter-depositing palladium onto the surface of a commercially available and inexpensive filtration membrane,” Zeng said.
The research was conducted at both Northern Illinois University and Argonne National Laboratory. The scientists also found that the speed of the sensors increases with decreasing thicknesses of the palladium nanowires. The sensors are 10 to 100 times faster than their counterparts made of a continuous palladium film of the same thickness.
“The superior performance of the ultra-small palladium nanowire network-based sensors demonstrates the novelty of the fabrication approach, which can be used to fabricate high-performance sensors for other gases,” said NIU Presidential Research Professor of Physics Zhili Xiao, leader of the research team and co-adviser to Zeng.
Xiao noted that Zeng’s exceptional contribution to the research is particularly impressive for a Ph.D. candidate. Zeng came to NIU in the fall of 2008 after earning her master’s degree from the University of Science and Technology Beijing. She is now a recipient of the NIU Nanoscience Fellowship, jointly supported by the university and Argonne.
“It is extremely competitive to publish an article in Nano Letters, which has a very high impact factor that is better even than the traditionally prestigious chemical and physical journals,” Xiao said. “We’re proud of Vivian’s achievements and grateful for her creativity and diligence.
“Nanoresearch is truly interdisciplinary,” Xiao added. “Chemists have undoubtedly demonstrated advantages in nanofabrication by utilizing methods of chemical synthesis to obtain extreme nanostructures, while physicists have strengths in exploration of new physical properties at the nanoscale. This research benefitted tremendously from Vivian’s expertise in chemistry. In fact, the substrates used to form the novel networks of palladium nanowires are common filtration members known to chemists.”
Provided by Northern Illinois University