Computer simulation strengthens link between climate change and release of subsea methane
(PhysOrg.com) -- A first-of-its-kind computer simulation that mirrors real-world observations of methane bubbling up from a seabed in the Arctic Ocean provides further evidence that warming oceans may unleash vast quantities of methane trapped in hydrate deposits buried beneath the seafloor.
A first-of-its-kind computer simulation that mirrors real-world observations of methane bubbling up from a seabed in the Arctic Ocean provides further evidence that warming oceans may unleash vast quantities of methane trapped in hydrate deposits buried beneath the seafloor. Methane is one of the most potent greenhouse gases.
The simulations, conducted by Lawrence Berkeley National Laboratory scientists, focus on oceanic gas hydrates, which are solid crystalline compounds in seafloor sediment. Methane molecules are lodged within the compounds’ icy latticework.
Scientists are concerned that climate change-induced ocean warming may cause oceanic hydrates to break apart, potentially releasing large amounts of methane into the atmosphere and further accelerating climate change.
Much more research is needed to understand this process, however, which is why Berkeley Lab scientists George Moridis and Matthew Reagan ran the largest-scale simulation ever conducted on the break up of oceanic gas hydrates.
Reagan will present their research today at the American Geophysical Union’s annual meeting in San Francisco. Their research is also published this week in the scientific journal Geophysical Research Letters.
They based their simulation on a seafloor off the coast of Spitsbergen, a Norwegian island in the Arctic Ocean where a recent scientific expedition observed more than 250 methane plumes.
The methane’s source isn’t known. But there is one intriguing clue: The seabed slopes upward as it approaches the island’s coastline, and the methane vents happen to occur at the depth where the water becomes too warm for methane hydrates to remain stable. Because of this, some scientists speculate that the plumes may be the first visible sign of the dissociation of methane hydrate deposits due to climate change.
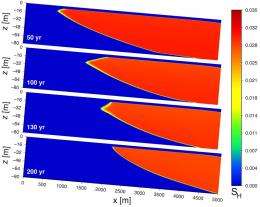
To help test this hypothesis, the Berkeley Lab scientists created a simulation that matches the seabed’s slope and depth over a five-kilometer, two-dimensional slice of seafloor. They cranked up the water temperature three degrees Celsius over a 100-year period, matching a temperature increase recorded in the waters off Spitsbergen since 1908. The ocean temperature was also held constant for another 100 years into the future.
They then watched what happened to methane hydrates buried in up to 300 meters of sediment.
As the water warmed, the boundary at which the hydrates remain stable crept deeper down the sloping seabed, dissociating the hydrates in the sediment and creating a receding line of gas plumes along the way. And when the simulation stopped at the water temperature currently found in the waters off Spitsbergen, this boundary line stalled near the depth where the methane vents were spotted by the expedition.
“Our simulations are consistent with what has been suggested based on the field observations: the methane is coming from the dissociation of shallow methane hydrates,” says Moridis of Berkeley Lab’s Earth Sciences Division.
Adds Reagan, “The location of the methane plumes match the receding gas hydrate stability zone in our simulation, which for us was the ’smoking gun’.”
The simulation uses a computer code developed at Berkeley Lab, called TOUGH+HYDRATE, which allows Moridis and Reagan to model the dissociation of hydrates and the migration of gas in the sediments. To model this system, they used 300,000 gridblocks to capture features as small as 25 centimeters.
“This is an extremely high-resolution simulation,” says Moridis. “And this gives us a very accurate description of the system, which is critical because hydrates are very heterogeneous.”
The simulation will help shore up what scientists know about the potential impact of methane hydrates on climate change, which remains hazy. Estimates vary widely over how much methane is trapped in oceanic hydrates. It’s also unclear how much methane will make it to the upper atmosphere, where it can contribute to global warming, as a result of oceanic hydrate dissociation. What is known is the physics of hydrate dissociation, and this offers a way to fill in knowledge gaps from the bottom up.
“We start with well-defined information about hydrates, and then simulate their behavior underneath the seafloor,” says Reagan. “Our simulations can then be coupled to ocean circulation and atmospheric models, which will eventually give us an estimate of the possible effect on global climate.”
More information: Read more about the computer simulation in a paper published in Geophysical Research Letters.
Provided by Lawrence Berkeley National Laboratory
















