New and improved right to the last drop
Biomolecules have become almost ubiquitous in laundry and personal care products. Biological washing powders usually contain enzymes to help digest stains while the latest shampoos and conditioners often use proteins to add shine and thickness to hair.
Now, researchers writing in the latest issue of Advanced Materials have developed a way to trap these biomolecules in tiny silica gel beads that could help get your clothes cleaner still by swelling to release the biomolecule at the appropriate time in the wash cycle. The gel beads could also extend the shelf life of countless consumer products so that they continue working well right to the last drop.
Bakul Dave and colleagues at Southern Illinois University Carbondale and Genencor International in Palo Alto, California, have come up with a "new and improved" way of making detergents and other cleaning and conditioning products that really does work. They used tiny beads of silica gel to trap biomolecules, such as enzymes known as alkaline proteases, which are commonly used in liquid detergents and washing powders to help remove proteins and other stains from dirty clothes.
The gel beads act as microscopic reservoirs for the biomolecules, releasing their contents only when the conditions are right in the wash. This means that the fragile enzymes, which can stop working after being exposed to heat, air and light for too long on the shelf, are protected from the outside world until they are needed in the washing machine. This means the product will continue to work as well as the manufacturer intended until the very last drop is poured from the bottle.
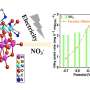
The microscopic gel beads are synthesised from the precursor bis[3-(trimethoxysilyl)propyl] ethylenediamine (enTMOS). The researchers demonstrated that these beads can swell and contract depending on the chemical conditions in which they find themselves. In concentrated form, the gel beads remain concentrated and tightly sealed. However, add water and the concentration drops and they are free to expand. It is this swelling process that is key to protecting the biomolecules until they are needed. The biomolecules can be trapped inside the beads during manufacture and will only be released promptly by adding water so that they remain isolated and protected until they are needed.
The team tested their swell gels as an additive to two different detergents: the first was a generic detergent ingredient, concentrated sodium dodecyl sulfate (SDS), while the second was a commercial laundry product with its enzyme content destroyed by heating to 90 °C for one hour. They then added the swell gels containing a test enzyme to each detergent.
The gels remained tight and contracted in the detergents, but as water was added, they began to swell and release their contents "into the wash". The researchers also tested shelf-life of the gels in detergent and found that very little enzyme leached out of the gel beads during storage–just about one percent from their swell gels. Also, they found that the enzyme in the swell gels was much more stable and was able to function even after heating to 80 °C as opposed to total loss of activity with a control experiment using a standard enzyme solution.
The swell gels have obvious applications in laundry detergents and fabric care applications, say the researchers. They add that the unique mechanism they have created with these materials which swell and contract depending on their environment bears a "rather striking resemblance" to the way living cells maintain water content and could provide biologists with a simple model for studying this process.
Citation: Bakul C. Dave, Osmoresponsive Glasses: Osmotically Triggered Volume Changes of Organosilica Sol-Gels as a Means for Controlled Release of Biomolecules, Advanced Materials 2006, 18, No. 15, doi: 10.1002/adma.200600852
Source: Advanced Materials