'Express lanes' for ions: By aligning carbon nanotubes in electrodes, researchers boost performance
Actuators are devices that convert electrical energy into mechanical energy, such as the battery-powered device inside a cell phone that causes the phone to vibrate. When this process is reversed -- when a device converts mechanical energy into electrical energy -- the device is called an energy harvester, and that electrical energy is often stored for future use. An example would be a device inside a pacemaker that converts mechanical energy created by the motion of a pair of breathing lungs into electrical energy that can be used to charge the pacemaker’s batteries.
Both devices typically contain electromechanical materials, such as electroactive polymers, that are made of chainlike molecules that change in size or shape when stimulated by an electric field. But their efficiency and speed depend on how quickly ions, or electrically charged particles, can move between electrodes, or the conductors that electric current passes through, to change the polymer’s size or shape. The faster ions can move between electrodes, the more ionic conductivity those electrodes will have and the more responsive the material will be to the electric field. Although these polymers usually contain nanoparticles that are randomly dispersed throughout the material to make it conductive, this slows ions by forcing them to travel in zigzag paths around the tiny particles.
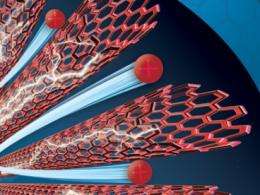
Recently, an MIT researcher collaborated with a team of electrical engineers from Pennsylvania State University to devise a new way for ions to travel more quickly between electrodes than they do in traditional polymers. Brian L. Wardle, associate professor of aeronautics and astronautics, and his colleagues designed electrodes containing aligned carbon nanotubes — tiny, hollow cylinders made of carbon atoms — to be used in an electroactive polymer. As they report in a paper to be published Oct. 8 in Advanced Functional Materials, this alignment created “express lanes” that enabled the ions to travel more quickly between electrodes. Specifically, the researchers estimate that the ionic conductivity of these electrodes is about an order of magnitude greater than that of electrodes in polymers that contain randomly dispersed nanoparticles.
Wardle and his colleagues, including Qiming M. Zhang, a professor of electrical engineering at Penn State, and lead author Sheng Liu, one of Zhang’s graduate students, demonstrated that the aligned carbon-nanotube electrodes can enhance ion performance in an actuator, meaning they could be optimized for applications like artificial muscles and robots.
The researchers state that the devices could be used as energy harvesters through a reverse-conversion process. There is tremendous interest in developing energy harvesters for large-scale applications, such as to create electrical energy from the motion of wind or ocean waves, Wardle says. The devices could also be used to power vast networks of microscopic sensors in hard-to-reach areas like underground pipes.
Composite creation
The researchers’ goal was to design a composite that could function as a superior electrode. By heating natural gas and exposing it to a metal catalyst, Wardle and several of his graduate students grew the electrically conductive carbon nanotubes and poured a polymer mixed in a solvent over them. Once the solvent evaporated, it left behind a solid, ion-porous composite containing both polymer and carbon nanotubes. The researchers then used this composite to create a structure comprised of one layer of pure polymer (to act as an insulator) containing both positive and negative ions sandwiched between two layers of the composite made of both polymer and carbon nanotubes (to act as electrodes).
To test the structure’s actuator abilities, the researchers applied a low-voltage electric field. This voltage caused ions to flow from one electrode layer to the other, which resulted in one side of the structure containing more ions. This ion imbalance generated enough pressure to cause the entire structure to bend, thereby creating mechanical energy. The experiment also revealed that the composite electrode design helped to minimize electrical resistance.
The researchers believe the same device could be used as an energy harvester if it is stressed mechanically, such as through compression. That’s because compression would make the ions move differently, which would cause an unbalanced electrical charge. This, in turn, would create a voltage difference and produce a flow of electricity.
Optimizing design
Yoseph Bar-Cohen, a senior research scientist at NASA’s Jet Propulsion Laboratory, says that the study demonstrates an improvement of ionic polymers. But he is curious about the response of the device over longer periods of time, noting that the current study was limited to only a 10-minute experiment.
As they develop these electrodes, Wardle and his collaborators are trying to determine an optimal design. Now that they have demonstrated how effective carbon nanotubes are for electrode efficiency, they are exploring certain details that might enable optimal performance, such as the spacing between the tiny tubes.
This story is republished courtesy of MIT News (web.mit.edu/newsoffice/), a popular site that covers news about MIT research, innovation and teaching.
Provided by Massachusetts Institute of Technology