In plants, small changes make big impact
(PhysOrg.com) -- You can’t see them or feel them, but right now countless biochemical interactions in your body affect your life in countless ways. These interactions are important because if they go poorly, really bad things can happen. In a simple and admittedly extreme example, poorly regulated protein interactions – interactions between molecules that are the main regulators of biochemical pathways inside cells – could result in unlimited cell division. That doesn’t sound so bad, unless you realize that there’s another name for it: cancer.
Dr. Steven Clouse studies these kinds of biochemical interactions, and the mechanisms behind them, in plants. Like humans and other animals, plants have messenger chemicals called hormones that, by binding to specific proteins, control the biochemical interactions that are essential for cellular functions like defense, growth and development, and many other things. When hormones bind to these proteins, it starts a cascade of interactions that result in changes to plant form and function.
A horticultural scientist and molecular biologist, Clouse specifically studies receptor kinases, the protein catalysts on cell surfaces that respond to the signals from hormones and other small molecules. There are more than 400 receptor kinases in the lab rat of plant studies, Arabidopsis thaliana, or mustard weed, and they initiate many diverse signaling pathways within the cell after binding hormones on the cell surface.
In one type of chemical interaction, a phosphate molecule will be added to a receptor kinase in response to hormone binding – the process is called phosphorylation – triggering other chemical interactions that lead to important functions like plant growth or stem elongation. Clouse also examines what happens when this process is altered or stifled.
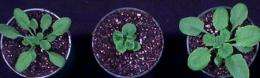
Think of phosphorylation as a regulatory on-off switch that can change cells in some way, Clouse says. When receptor kinases are not correctly phosphorylated, plants can’t grow properly. The result: short-leafed, defective-looking plants that can’t reproduce.
“These hormones – called brassinosteroids – are essential for plant growth; the receptor kinases that bind them are essential for plant growth; and resulting phosphorylation is also essential for plant growth,” Clouse says.
Clouse and Dr. Michael Goshe, NC State professor of molecular and structural biochemistry, and Dr. Steven Huber, a former NC State professor who now works at the University of Illinois, study two particularly important receptor kinases required for brassinosteroid signaling, BRI1 and BAK1, that are key to cell division, stem elongation, and other growth and development functions in plants.
In the past five years, the findings of this team of faculty – along with their postdoctoral scientists and graduate students – on the molecular mechanisms behind hormone-protein interactions have appeared in a number of high-octane scientific journals. One paper in the journal Plant Cell, in 2005, identified specific phosphorylation sites for BRI1, using a combination of genetics, biochemistry and mass spectrometry.
In 2008, a paper in Developmental Cell showed the order of phosphorylation in BRI1 and BAK1 interactions and how it affects plant growth. It also provided the first detailed model of how plant receptor kinases bind with each other and activate each other, and compared this mechanism to the better-known animal models.
More recently, Clouse and colleagues published two papers in Proceedings of the National Academy of Sciences that showed even more details about specific phosphorylation sites.
These phosphorylation sites are important because they can be altered in ways that positively affect agricultural traits. Clouse says a research group overseas altered important BRI1 phosphorylation sites in rice plants and attained up to 30 percent higher yields from the affected plants.
Clouse’s National Science Foundation funding has come through the Arabidopsis 2010 project, which aims to discover the functions of all 30,000 Arabidopisis genes by 2010.
In 2004, Clouse and colleagues received $2 million over four years to study 223 receptor kinases – like BRI1 and BAK1 – and examine how they phosphorylate.
This summer, the team received a competitive renewal for $2.5 million over the next four years. The grant has a number of goals, two of which are to examine 50 individual receptor kinases by proteomic approaches – essentially looking at the broad spectrum of proteins at the same time. They’ll discover what binds to these receptor kinases and figure out how they interact with other receptor kinases and proteins. They’ll also closely examine 12 of these 50 receptor kinases and discover their function.
In the end, “Small changes at individual phosphorylation sites affect plant growth,” Clouse says. “Understanding the mechanisms behind how plants grow and defend themselves has many practical applications in agriculture.”
Provided by North Carolina State University

















