Researchers develop MRSA-killing paint

Building on an enzyme found in nature, researchers at Rensselaer Polytechnic Institute have created a nanoscale coating for surgical equipment, hospital walls, and other surfaces which safely eradicates methicillin resistant Staphylococcus aureus (MRSA), the bacteria responsible for antibiotic resistant infections.
"We're building on nature," said Jonathan S. Dordick, the Howard P. Isermann Professor of Chemical and Biological Engineering, and director of Rensselaer's Center for Biotechnology & Interdisciplinary Studies. "Here we have a system where the surface contains an enzyme that is safe to handle, doesn't appear to lead to resistance, doesn't leach into the environment, and doesn't clog up with cell debris. The MRSA bacteria come in contact with the surface, and they're killed."
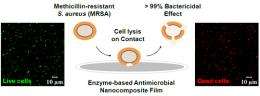
In tests, 100 percent of MRSA in solution were killed within 20 minutes of contact with a surface painted with latex paint laced with the coating.
The new coating marries carbon nanotubes with lysostaphin, a naturally occurring enzyme used by non-pathogenic strains of Staph bacteria to defend against Staphylococcus aureus, including MRSA. The resulting nanotube-enzyme "conjugate" can be mixed with any number of surface finishes — in tests, it was mixed with ordinary latex house paint.
Unlike other antimicrobial coatings, it is toxic only to MRSA, does not rely on antibiotics, and does not leach chemicals into the environment or become clogged over time. It can be washed repeatedly without losing effectiveness and has a dry storage shelf life of up to six months.
The research, led by Dordick and Ravi Kane, a professor in the Department of Chemical and Biological Engineering at Rensselaer, along with collaboration from Dennis W. Metzger at Albany Medical College, and Ravi Pangule, a chemical engineering graduate student on the project, has been published in the July edition of the journal ACS Nano, published by the American Chemical Society.
Dordick said the nanotube-enzyme coating builds on several years of previous work embedding enzymes into polymers. In previous studies, Dordick and Kane discovered that enzymes attached to carbon nanotubes were more stable and more densely packed when embedded into polymers than enzymes alone.
"If we put an enzyme directly in a coating (such as paint) it will slowly pop out," Kane said. "We wanted to create a stabilizing environment, and the nanotubes allow us to do that."
Having established the basics of embedding enzymes into polymers, they turned their attention to practical applications.
"We asked ourselves — were there examples in nature where enzymes can be exploited that have activity against bacteria?" Dordick said. The answer was yes and the team quickly focused on lysostaphin, an enzyme secreted by non-pathogenic Staph strains, harmless to humans and other organisms, capable of killing Staphylococcus aureus, including MRSA, and commercially available.
"It's very effective. If you put a tiny amount of lysostaphin in a solution with Staphylococcus aureus, you'll see the bacteria die almost immediately," Kane said.
Lysostaphin works by first attaching itself to the bacterial cell wall and then slicing open the cell wall (the enzyme's name derives from the Greek "lysis" meaning "to loosen or release").
"Lysostaphin is exceptionally selective," Dordick said. "It doesn't work against other bacteria and it is not toxic to human cells."
The enzyme is attached to the carbon nanotube with a short flexible polymer link, which improves its ability to reach the MRSA bacteria, said Kane.
"The more the lysostaphin is able to move around, the more it is able to function." Dordick said.
They successfully tested the resulting nanotube-enzyme conjugate at Albany Medical College, where Metzger maintains strains of MRSA.
"At the end of the day we have a very selective agent that can be used in a wide range of environments — paints, coating, medical instruments, door knobs, surgical masks — and it's active and it's stable," Kane said. "It's ready to use when you're ready to use it."
The nanotube-enzyme approach is likely to prove superior to previous attempts at antimicrobial agents, which fall into two categories: coatings that release biocides, or coatings that "spear" bacteria.
Coatings that release biocides — which work in a manner similar to marine anti-fouling paint — pose harmful side-effects and lose effectiveness over time as their active ingredient leaches into the environment.
Coatings that spear bacteria — using amphipatic polycations and antimicrobial peptides — tend to clog, also losing effectiveness.
The nanotube-lysostaphin coating does neither, said Dordick.
"We spent quite a bit of time demonstrating that the enzyme did not come out of the paint during the antibacterial experiments. Indeed, it was surprising that the enzyme worked as well as it did while remaining embedded near the surface of the paint," Dordick said.
The enzyme's slicing or "lytic" action also means that bacterial cell contents disperse, or can be removed by rinsing or washing the surface.
Kane also said MRSA are unlikely to develop resistance to a naturally occurring enzyme.
"Lysostaphin has evolved over hundreds of millions of years to be very difficult for Staphylococcus aureus to resist," Kane said. "It's an interesting mechanism that these enzymes use that we take advantage of."
Provided by Rensselaer Polytechnic Institute
















