Unexpected Hydrides Become Stable Metals at Pressure Near One Quarter Required to Metalize Pure Hydrogen Alone
(PhysOrg.com) -- From detailed assessments of electronic structure, researchers at the University at Buffalo, Cornell University, Stony Brook University and Moscow State University discovered that unexpected hydrides violating standard valence rules, such as LiH6 and LiH8, become stable metals at a pressure approximately one quarter of that required to metalize pure hydrogen itself; findings that were published in an October 5, 2009 early edition of the Proceedings of the National Academy of Sciences.
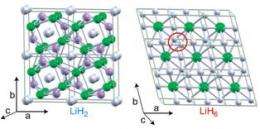
The paper, entitled "A little bit of lithium does a lot for hydrogen," presents the first prediction of stable LiHn hydrides (LiH2, LiH6, LiH8). These hypothetical materials demonstrate that nontraditional stoichiometries can considerably expand the view of chemical bonding already under moderate pressure.
Metallic hydrogen, believed to be stable at high pressures, is theorized to be a superconductor at record high temperatures of at least a few hundred Kelvin (room temperature or higher). Due to its high (100%) hydrogen content and high density it is the ultimate energy storage material - if it can be synthesized in large quantities and subsequently brought to ambient conditions in the same metallic form.
For decades, researchers at the top research institutions around the world have predicted exotic properties for metallic hydrogen, but no credible reports of experimental synthesis of solid metallic hydrogen ever appeared because of two primary obstacles. First, metallization of hydrogen requires pressures of about four million atmospheres, which was out of reach of static compression techniques. Extreme pressures, even if they could be reached, imply that only tiny amounts of the material can be prepared, which would be of little practical use. Second, the recovery of this high pressure material to ambient pressure will be almost certainly problematic.
Work of Eva Zurek, assistant professor at the University at Buffalo, her former Cornell University colleagues Roald Hoffmann and Neil W. Ashcroft, in collaboration with Professor Artem R. Oganov and his colleague Andiry O. Lyakhov at Stony Brook University, offers surprising new optimism.
"Synthesis of metallic hydrogen has long been a dream of physicists. A dream that is now one big step closer, thanks to this theoretical work," says Professor Oganov.
"There are fundamental reasons to be excited about this form of metallic hydrogen. Light nucleus of hydrogen behaves like a quantum particle-wave, making it possible that there will be altogether new states of matter, simultaneously superconducting and superfluid."
To uncover these findings, Zurek tapped the powerful computational methods developed by Oganov and Lyakhov - methods which allow one to predict the structure and composition of new stable compounds before they are synthesized in the lab. What Zurek found was that, while LiH is a simple and well-known material at normal conditions, very unusual chemistry appears at pressures above one million atmospheres.
Unexpected hydrogen-rich metallic compounds, such as LiH2, LiH6 and LiH8 become stable. Many of their properties would be similar to those of the long-sought metallic hydrogen, but conditions of synthesis can be readily achieved in the lab. The study also shows a way to prepare metallic almost-hydrogen for possible practical use.
"Finding elements that would form such compounds at still lower pressures is now the most realistic solution to the metallic hydrogen problem and opens the door to a world of new chemistry, where little can be anticipated with traditional chemical concepts - no one would have expected LiH8, LiH6 or LiH2 to be stable compounds," says Oganov. "It is possible that new important chemical rules will be found along this exploratory path. And who knows, maybe one day this will lead us to room-temperature superconductivity and new twists in the search for hydrogen storage materials."
More information: PNAS article link: www.pnas.org/content/early/200 … 262106.full.pdf+html
Provided by Stony Brook University (news : web)



















