Scientists follow live infection by food-poisoning bacteria Listeria
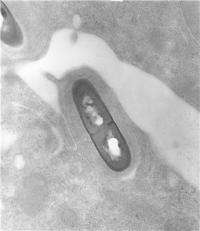
Scientists in Portugal and France managed to follow the patterns of gene expression in food-poisoning bacteria Listeria monocytogenes (L. monocytogenes) live during infection for the first time. The work about to be published in PLoS Pathogens shows how the bacterial genome shifts to better adapt to infection by activating genes involved in virulence and subversion of the host defences, as well as adaptation to the host conditions. This is the first time that the molecular interactions between L. monocytogenes and its host, as they occur during the different steps of infection, are followed in real time paving the way, not only to the development of new therapies against this potentially lethal bacterium, but also for the study of other pathogen/host interactions.
L. monocytogenes exists everywhere in nature what is not a problem for individuals with healthy immune systems capable of keep the bacteria at bay, but a real danger for immunocompromised or pregnant hosts, in which case Listeriosis (the infection) can result in a mortality rate as high as 30%. The disease, which normally occurs after ingestion of contaminated food, starts in the digestive system with the bacteria moving next to the liver and spleen. There, if not controlled by the immune system, will continue to multiply and eventually lead to septicaemia or, if it migrates into the brain, to meningitis as well being able to induce miscarriages in the case of pregnant women after infection of the foetus. Although the disease is relatively rare and there are antibiotics, these are not particularly efficient - probably because of the bacteria capability to hide inside the cells - as revealed in the high mortality rate found among susceptible individuals. This makes Listeriosis an important public health problem and not only in the developing world - where AIDS and poor nutrition create high numbers of vulnerable individuals - but also in countries such as the US where about 1600 new cases and 500 deaths are reported every year.
To understand better the infection Ana Camejo, Didier Cabanes and colleagues from the Institute for Molecular and Cellular Biology at Oporto University, Portugal and the Pasteur Institute in Paris, France made use of a powerful approach called DNA array, which allows to follow gene expression by detecting the RNA molecules being produced at a certain time point. In fact, during the process of gene expression the information contained in the DNA is converted into RNA molecules that are then read by the body machinery to produce the corresponding proteins. DNA arrays are plates of spots with attached DNA fragments which bind and detect their corresponding RNA, and by detecting the RNA present in a cell they effectively identify the genes that are active at a certain time point.
In the case of L. monocytogenes it is known that about 19% of its genome is differentially expressed during infection and to identify those genes is key to understand such events as the bacteria capacity to cross the intestinal, brain and placenta barriers or to infect the immune system cells without being targeted by it, all crucial for the bacteria high lethality.
To discover this Camejo, Cabanes and colleagues compared RNA molecules collected at different times (24, 48 and 72 hours) after infection from bacteria infecting mice, with the RNA from bacteria growing in laboratory, the idea being, that those genes only activated in the bacteria infecting mice but not when in laboratory would be linked to the infectious process. And in fact, it was found that about 20% f the bacterial genome was differentially expressed during Listeriosis with most of the activated genes linked to subversion of the host’s defence - such as for example the production of a negative charge in the bacterial wall to repel the host anti-microbial peptides -, resistance to the stress induced by the host defences and several adaptations to the new environment.
Camejo and colleagues also found that these infection-linked genes were controlled by a complex network coordinated by two major regulators called PrfA and SigB.
Finally, the researchers were able to identify several new virulence factors, which are bacterial proteins that induce the disease in the host.
Camejo, Cabanes and colleagues’ work reveals how DNA arrays can give a detailed image of the molecular interactions between pathogen and host as they are occurring in the host in real time and how this can be a powerful tool to understand infection as demonstrated by the discovery of several new virulence factors for Listeria. This information can now be used in the development of more efficient interventions against the disease, particularly in more susceptible individuals but also paves the way for the use of the technique in other infectious where the knowledge of the host/pathogen interaction is still scarce.
More information: Public Library of Science (PLoS) Pathogens -to be published on the 28th of May 2009; “In Vivo Transcriptional Profiling of Listeria monocytogenes and Mutagenesis Identify New Virulence Factors Involved in Infection”
Source: Ciência Viva - Agência Nacional para a Cultura Científica e Tecnológica















