Materials' crystal properties illuminated by mathematical 'lighthouse'
A deeper fundamental understanding of complex materials may now be possible, thanks to a pair of Princeton scientists who have uncovered a new insight into how crystals form.
The researchers' findings reveal a previously unknown mathematical relationship between the different arrangements that interacting particles can take while freezing. The discovery could give scientists insight into the essential behaviors of materials such as polymers, which are the basis of plastics.
Molecules in a material cooled to absolute zero can take on a multitude of different configurations. Historically, scientists' difficulty with identifying crystallized molecules' spatial arrangements from this high number of possible configurations has blocked theoretical efforts to understand these materials' qualities, but the new findings could offer the tool that science needs.
"We believe our 'duality relations' will be a useful theoretical tool to understand how individual particles come together to form a crystal," said Salvatore Torquato, a professor of chemistry who co-wrote the paper with senior chemist Frank Stillinger. "If we can tune the interactions among particles that form a crystal, we might be able to create materials that respond to light or mechanical stress in novel ways."
A material that maintains its exact size and shape through extremes in temperature, for example, might be valuable in the manufacture of orbiting space telescopes, whose mirrors need to retain their shape as they pass from sunlight into the Earth's shadow.
A crystal is the state of matter that is easiest to analyze because its frozen molecules are motionless and often regularly organized. A crystal's properties -- its ability to bend light, for example -- generally reveal valuable information about how its constituent molecules will behave at higher temperatures, such as when they become a liquid.
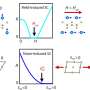
The challenge is that many complex materials can crystallize into a multitude of different structures. When a substance is cooled to nearly absolute zero, and it can take on an enormously large number of possible "ground states" -- the term for the molecular arrangement with the lowest possible energy. Scientists seek to determine the true ground state because it provides a fundamental understanding of matter in the solid state and its possible uses. However, determining which molecular pattern is the true ground state requires mathematical proof that is hard to come by.
"We resort to approximations," said Christos Likos, a professor of theoretical physics at the University of Dusseldorf in Germany. "They help us produce meaningful results sometimes, but we need to have a lighthouse occasionally to show us we're on the right path. Such lighthouses are rare in this business, but Sal and Frank have found one."
Torquato and Stillinger's findings explore particles' behavior as they attract and repel each other over varying distances. By analyzing this behavior, the scientists were able to conceive a precise mathematical correspondence -- called duality relations -- between possible arrangements of particles. The work will enable the researchers to draw important conclusions about how particles at very low temperatures interact over great distances, a situation that is very difficult to treat theoretically.
"Once ground states can be determined and controlled with certainty, scientists might create materials with properties virtually unknown in nature," Torquato said.
The Department of Energy funded the team's research, which appears in the Jan. 16 edition of the journal Physical Review Letters.
Source: Princeton University