Slavery carried bilharzia parasites from West Africa to the Caribbean, genomics confirms
The bilharzia-causing parasite, Schistosoma mansoni, first infected humans as they fished in lakes in East Africa and was spread, first to West Africa and then to the New World by slave traders in 16th-19th Centuries, genomics reveals.
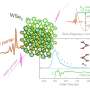
Today (16 February) in Scientific Reports, Wellcome Trust Sanger Institute, Imperial College London and Royal Veterinary College scientists used the full DNA sequences of Schistosoma mansoni parasites from Africa and the French Caribbean to discover the fluke's origins, map its historic transmission and identify the secrets of its success. Their findings show how the global slave trade transported the disease from Senegal and Cameroon to Guadeloupe. Further genomic comparison with a closely related schistosome species that infects rodents reveals how the parasite has adapted to infecting human beings.
Schistosoma mansoni is a blood fluke (flatworm) that infects more than 250 million people worldwide and causes more than 11,000 deaths each year. Six years ago the Sanger Institute published the parasite's first full DNA sequence (genome); this latest study used that 'genetic map' to construct and compare the genomes of S. mansoni parasites gathered from across Africa and the New World, the majority of which were held at the Schistosomiasis Collection in the Natural History Museum, London.
By analysing the differences between the human-infecting S. mansoni and its close relative, the rodent-infecting S. rodhaini, the scientists calculated that the two species evolved from a common ancestor approximately 107,000 to 148,000 years ago in East Africa. This finding suggests that the species is much 'younger' than previously thought.
"The timing of the separation of the two species coincidences with the first archaeological evidence of fishing in Africa," explains Thomas Crellen, first author of the study from Imperial College London, the Sanger Institute and the Royal Veterinary College London. "The parasite develops in freshwater and infects people by burrowing through their skin. The introduction of fishing would have meant that people spent more time in the water, greatly increasing their chances of being infected."
Analysing the differences between genomes from different locations also revealed the darker side of human history.
"Comparing the S. mansoni genomes suggests that flukes in West Africa split from their Caribbean counterparts at some point between 1117AD and 1742AD, which overlaps with the time of the 16th-19th Century Atlantic Slave Trade," says Professor Joanne Webster from Imperial College London and the Royal Veterinary College. "During this period more than 22,000 African people were transported from West Africa to Guadeloupe by French slave ships, and the fluke was carried with them."
Comparing the genomes of S. mansoni with S. rodhaini also revealed the genetic variations that have been positively selected over time in the human-infecting fluke and have been "fixed" into its DNA. It is likely that these variations are the evolutionary adaptations that have occurred to enable the fluke to successfully tunnel into, and thrive within, human beings.
"When we looked for the differences between human-infecting S. mansoni DNA and its rodent infecting cousin S. rodhaini, we found two important variations. We found that changes to two genes in S. mansoni's DNA - VAL21 and an elastase gene -appear to be important in allowing the fluke to enter and live in humans," says Dr James Cotton, senior author of the study from the Sanger Institute. "VAL genes produce proteins that cause allergic responses, so it is possible that the variation in VAL21 helps the fluke to hide from our immune systems. The elastase gene helps the parasite to burrow in to the body, by breaking down elastin - a major component of human skin."
It is hoped that exploring the genetic makeup of the fluke it will be possible to discover more about the processes the parasite relies on to infect humans and offer new opportunities to develop preventive and therapeutic interventions.
More information: Thomas Crellen et al. Whole genome resequencing of the human parasite Schistosoma mansoni reveals population history and effects of selection, Scientific Reports (2016). DOI: 10.1038/srep20954
Journal information: Scientific Reports
Provided by Wellcome Trust Sanger Institute