Scientists blueprint tiny cellular 'nanomachine'
Scientists have drawn up molecular blueprints of a tiny cellular 'nanomachine', whose evolution is an extraordinary feat of nature, by using one of the brightest X-ray sources on Earth.
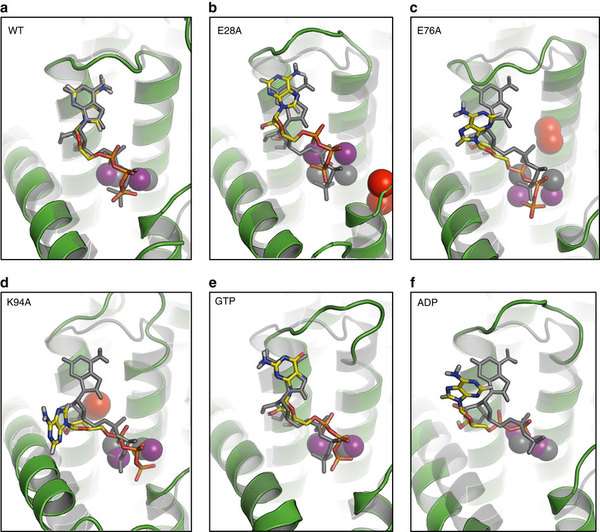
The scientists produced the structural map of this nanomachine – diacylglycerol kinase – by using a "hit and run" crystallography technique.
In doing so, they have been able to understand how the tiny enzyme performs critical cellular duties – answering questions that have been on the table for over 50 years about this 'paradigmatic protein'.
Kinases are key players in metabolism, cell signalling, protein regulation, cellular transport, secretory processes, and many other cellular pathways that allow us to function healthily. They coordinate the transfer of energy from certain molecules to specific substrates, affecting their activity, reactivity, and ability to bind other molecules.
Diacylglycerol kinase, the focus of this study, plays a role in bacterial cell wall synthesis. It is a small, integral membrane enzyme that coordinates a particularly complex reaction: its lipid substrate is hydrophobic (repelled by water) and resides in cell membranes, while its co-substrate, ATP, is entirely water soluble.
How it does this had remained a mystery for decades, but the newly produced blueprints have answered these questions.
"How this diminutive nanomachine, less than 10 nm tall, brings these two disparate substrates together at the membrane interface for reaction is revealed in a molecularly detailed crystal structure. It is the smallest known kinase, and seeing its form with crystal clarity is now helping us to answer questions that formed from over 50 years of work on this paradigmatic protein," said Professor of Membrane Structural and Functional Biology, in the School of Biochemistry and Immunology at Trinity College Dublin, Martin Caffrey.
Figuring out how this tiny machine works at the molecular level was enormously facilitated by our use of one of the brightest X-ray sources on Earth, the X-ray free-electron laser at the Stanford Linear Accelerator Center.
Professor Caffrey added: "This instrument produces bursts of X-rays just femtoseconds (a quad-trillionth of a second) long. With these short bursts we were able to obtain structural information about the enzyme before it vaporized through radiation damage in what I tritely refer to as 'Hit and Run' serial crystallography."
In the future, the scientists hope to extend their free-electron laser work to make 'X-ray movies' of this remarkable nanomachine, so as to watch how it 'does chemistry' in atomic detail in real time.
The article describing the work was today published in the leading journal Nature Communications.
More information: Dianfan Li et al. Ternary structure reveals mechanism of a membrane diacylglycerol kinase, Nature Communications (2015). DOI: 10.1038/ncomms10140
Journal information: Nature Communications
Provided by Trinity College Dublin