Arctic cod inspire new way to help hospitals keep blood on ice
Antifreeze proteins from fish living in icy seas have inspired a new way to freeze blood which could one day increase the precious stocks available for medical procedures.
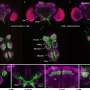
University of Warwick researchers have found a new application for a common polymer which allows blood cells to better survive being stored at freezing temperatures. The study is published in the journal Nature Communications.
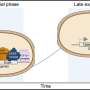
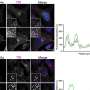
Polyvinyl alcohol – which mimics antifreeze properties found in cold-acclimatised fish like arctic cod - works by inhibiting the growth of ice crystals during thawing which would otherwise damage the blood cells and make them unusable in medical settings.
Currently in the UK, blood is stored by refrigeration – but it can only be kept this way for up to one month so hospitals are dependent on a constant supply of donors to keep blood banks topped up.
These stocks can fall at certain times of the year – for example over Christmas or during major sporting events – which is why freezing is an attractive option.
Many other donor cells also need to be cryopreserved, including bone marrow for leukaemia patients. The need for technologies such as one developed at the University of Warwick is expected to grow in importance with the growth of biobanks and the emergence of more stem cell therapies.
Some countries, including the US, already use blood cryopreservation but the current method requires the addition of large quantities of organic solvent, with every litre of blood requiring up to a litre of solvent to prevent the formation and growth of ice crystals which can kill the cells.
The organic solvent still has to be removed before the blood is used to save lives in a process which can take several days, whereas emergency transfusions require rapid availability.
The new system of freezing described by the University of Warwick researchers requires an additive of only 0.1 per cent of the volume of the blood.
Also, it doesn't need to be removed once the blood is defrosted so can be put to use rapidly.
The scientists believe the discovery offers a broad range of possible applications, from in vitro scientific research through to stem cell therapies in the clinic.
Along with Warwick Ventures, the University's technology commercialisation company, the scientists are looking to partner this technology with commercial organisations that have the expertise to develop such applications.
Dr Matthew Gibson from the Department of Chemistry at the University of Warwick said: "We know that certain types of fish survive perfectly well in sub-zero sea temperatures without their blood freezing.
"We used this as a starting point to search for synthetic substances which reflect what nature already does so well.
"On closer examination it turns out that polyvinyl alcohol – which is actually a derivative of wood glue - mimics the properties of the antifreeze proteins found in these kinds of fish.
"Polyvinyl alcohol has three things in its favour when applied to freezing blood.
"Firstly it reduces the growth of ice crystals during thawing, secondly it reduces the need for organic solvents – and crucially it reduces the time between defrosting and having transfusion-ready blood by eliminating the need to remove solvent.
"Although we still need to run further tests, this new method looks very promising in terms of vastly extending the shelf life of blood stored for medical procedures and therefore preventing dangerous dips in blood availability at certain times of the year.
"In addition to these benefits for blood we are excited by the scope for other applications including cell-based therapeutics for patients with rare and serious diseases including certain cancers and neurological conditions"
More information: "Synthetic Polymers Enable Non-Vitreous Cellular Cryopreservation by Reducing Ice Crystal Growth During Thawing." Robert C. Deller Manu Vatish Daniel A. Mitchell Matthew I. Gibson. Nature Communications 5, Article number: 3244 DOI: 10.1038/ncomms4244 Received 13 June 2013 Accepted 13 January 2014 Published 03 February 2014
Journal information: Nature Communications
Provided by University of Warwick