Researchers unveil method for creating 're-specified' stem cells for disease modeling
In a paper in Cell Stem Cell, a team led by researchers in the Boston Children's Hospital's Stem Cell Transplantation Program reports a new approach for turning induced pluripotent stem cells (iPSCs) into hematopoietic stem and progenitor cells for in vivo disease modeling.
With this strategy—which they call re-specification—the team, including Sergei Doulatov, PhD, and George Daley, MD, PhD, of Boston Children's, may have overcome technical barriers to generating blood disease-specific animal models from the thousands of iPSC cell lines now sitting in laboratory freezers around the world.
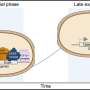
The main advantage of the technique lies in the raw material. The research team started with iPSCs that had already been directed to grow into myeloid progenitors, which are more closely related to the desired blood progenitors than skin or other fully differentiated cell types commonly used in stem cell experiments.
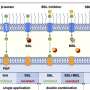
The researchers then used a select set of transcription factors to turn back the molecular clock just a little on these committed myeloid cells, turning them into blood progenitors that readily engrafted and differentiated when transplanted into mice.
The re-specification technique could help generate the large number of engraftable cells needed to create animal models from iPSCs generated from human patients suffering a range of blood disorders, such as anemias, thalassemia or sickle cell disease.
Journal information: Cell Stem Cell
Provided by Children's Hospital Boston