Clues to foam formation could help find oil
Blowing bubbles in the backyard is one thing and quite another when searching for oil. That distinction is at the root of new research by Rice University scientists who describe in greater detail than ever precisely how those bubbles form, evolve and act.
A new study led by Rice chemical and biomolecular engineer Sibani Lisa Biswal and published in the journal Soft Matter describes two previously unknown ways that bubbles form in foam.
The work should be of interest to those who make and use foam for a variety of reasons, from shaving cream to insulation. But it may be of primary importance to companies trying to extract every possible drop of oil from a reservoir by using volumes of thick foam to displace it.
Biswal and her team used microfluidic devices and high-speed imaging to capture images of how bubbles transform as they pass through tight spaces like those found in permeable rock deep underground. They discovered mechanisms that should help engineers understand how foam can be manipulated for specific tasks.
"In the classic descriptions of bubble formation, there's what we call snap-off, lamella division and leave-behind," Biswal said. Snap-off bubbles are created when liquid accumulates by capillary action in a narrow section of a pore and forms a liquid slug separating two bubbles. A lamella division bubble happens when the lamella (a thin film of liquid) moves through a branch in the flow path and becomes two lamella. Leave-behind happens when a gas enters two adjoining, parallel pores and the liquid between the two pores thin down to a lamella.
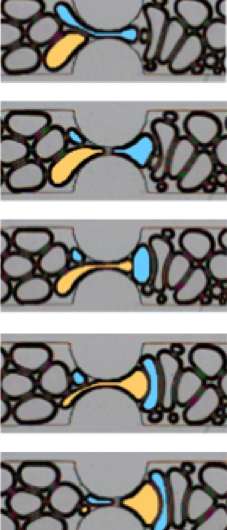
In the newly observed bubble-making processes, which she calls "pinch-off" behaviors, the bubbles form before gas passes through the constriction, not after.

"No one has seen these mechanisms," she said. In one pinch-off, a bubble caught between a neighboring bubble and the wall would split as it entered the channel. In the second, she said, "We found neighboring bubbles that are basically karate-chopping a third one as it tries to go through."
The smaller the bubbles in the foam, the better it may serve enhanced oil recovery, said George Hirasaki, a Rice research professor of chemical and biomolecular engineering and co-author of the paper.
"We're trying to understand how foam behaves in porous media because it is a way of making gas act like a more viscous fluid," he said. "Normally, gas has very low viscosity and it tends to flow through rock and not displace oil and water. Once it finds a path, usually along the top of a reservoir, the rest of the gas tends to follow.
"If there were some way to make gas act more like a liquid, to make it more viscous, then it would contact much more of the reservoir and would push the fluids out," Hirasaki said.
Ideally, foam would pack the channels inside high-permeable regions and force pressure to flow through rocks with low permeability, flushing out the hard-to-get oil often trapped there.
The Biswal lab built devices that mimic what happens in porous rock, squeezing mixtures of gas and surfactant through 20 micrometer-wide channels. They filmed what happened under a range of pressures at either end of the channel at 10,000 frames per second.
"Normally we work in rock samples or sand packs and we measure the pressure drop," Hirasaki said. "It's hard to see what's happening at the pore scale. But with the micromodel, we can see it with our own eyes – or with the camera's eye."
"We want to offer the oil industry more mobility control," Biswal said. "What we mean by that is the ability to drive fluids through areas that vary in their permeability. We want fluids to move through the entire path, not just the path of least resistance."
Lead authors are Rice alumna Rachel Liontas, currently a graduate student at Caltech, and former graduate student Kun Ma, currently a reservoir engineer at Total E&P USA. Biswal is an associate professor of chemical and biomolecular engineering.
More information: pubs.rsc.org/en/content/articl … m51605a#!divAbstract
Journal information: Soft Matter
Provided by Rice University