September 17, 2013 report
Criticality in morphogenesis
(Phys.org) —In many regards, a brief time-lapse video can teach more about embryonic development than any amount of reading. It is hard not to be impressed how a repeatable form reliably emerges despite considerable variation in both genes and environment. While it had been hoped that concepts borrowed from statistical mechanics or the ideas of self-organized criticality could help to create some kind of physics-based theory of development, much of what has been done lies only at the level of metaphor. In a paper just released to ArXiv, William Bialek and his colleagues from Princeton University, have taken their search for the signature of criticality in a more specific direction. They looked at a particular set of transcription factors in Drosophila embryos which control spatiotemporal development. By analyzing fluctuations in the expression levels of these so-called gap genes, they found evidence for critical tuning in this particular network.
Gap genes are of particular interest in Drosophila where they serve as "maternal effect genes." Here they directly transduce information from the mother to the embryo through residual mRNA and other cell components. Gap genes can control development through specific patterns generated in part by cross regulation and mutual inhibition. While it is possible that the phenotype, or appearance, of the organism arises from the actions of attractors in the gap gene dynamical system, the timing of critical events in embryogenesis need not necessarily correspond to critical points in gene expression.
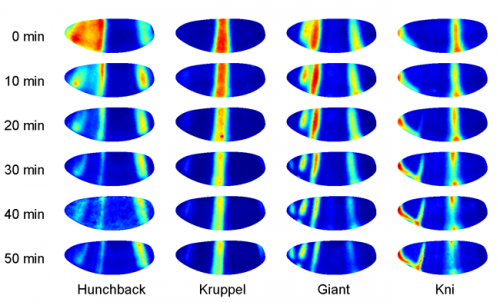
In the middle region of the embryo two gap genes, Hb and Kr are expressed at significant levels around the time period specific to the 14th nuclear division cycle. One prediction of criticality is that fast dynamics in expression levels should occur in directions along the embryo where the fluctuations are small, and slow dynamics (variations over time periods of an hour) should occur in the directions where their are large fluctuations.
To evaluate this prediction, the researchers used measurements of the protein concentrations of the gene products rather than the direct mRNA expression levels. They report that this immunostaining method gives protein concentrations that are accurate to within 3% of the expression levels. Their results they obtained were in good agreement with these predictions for criticality. They also looked at the correlations between gap gene fluctuations at each position in the embryo. For the "crossing regions" where the levels were transiently the same, the correlation coefficient was found to approach -1, indicating perfect anticorrelation as expected at criticality.
The authors suggest that the gap gene network must be finely tuned in order to generate the observed large differences in the timescales for variation in different directions. Similar tuning should be required to maintain the observed correlations over large distances. At the same time however, there must also be a certain level of robustness to details like sequence variation and alterations in the environment in which the embryo develops.
Within this particular study, only a couple components of transcription were even evaluated. The authors note that transcriptional regulation is just one context where criticality can operate in biological systems. A similar approach may be taken to look at other systems like for example, neural networks critically poised between quiescence and seizure, or the group behavior of flocks schools. The idea of "avalanches" of all sizes occurring in the system might even be applied to ideas of punctuated equilibrium in evolutionary biology as whole. Looking for deeper principles in biology is perhaps somewhat of a subjective business, but starting with well defined examples backed with abundant data is a great place to start.
More information: Morphogenesis at criticality? arXiv:1309.2614 [q-bio.MN] arxiv.org/abs/1309.2614
Abstract
Spatial patterns in the early fruit fly embryo emerge from a network of interactions among transcription factors, the gap genes, driven by maternal inputs. Such networks can exhibit many qualitatively different behaviors, separated by critical surfaces. At criticality, we should observe strong correlations in the fluctuations of different genes around their mean expression levels, a slowing of the dynamics along some but not all directions in the space of possible expression levels, correlations of expression fluctuations over long distances in the embryo, and departures from a Gaussian distribution of these fluctuations. Analysis of recent experiments on the gap genes shows that all these signatures are observed, and that the different signatures are related in ways predicted by theory. While there might be other explanations for these individual phenomena, the confluence of evidence suggests that this genetic network is tuned to criticality.
Journal information: arXiv
© 2013 Phys.org