Gold probes may offer valuable insight into cancer
(Phys.org) —Nanoprobes made from gold could be used to predict people's cancer risk – and the effectiveness of treatments, following research by University of Strathclyde academics.
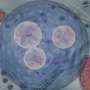
The nanoprobes could allow scientists to study cancer cells in minute detail – using a highly-sensitive imaging technique known as FRET microscopy – with the aim of identifying tumour-causing properties. The nanoprobes could also be used to measure how effective medicines are, at a sub-cellular level of detail, while another application could be the identification of contaminants in food and water supplies.
Dr Yu Chen, of the University's Department of Physics, said: "The technology could allow the simultaneous detection of multiple types of RNA related to cancer, which would then raise the possibility of scientists eventually being able to screen patients, in order to predict their risk of developing disease. By allowing us to see what is happening inside cells, we also hope this research will also lead to the development of techniques to study the efficacy of drugs."
Co-worker Professor David Birch, also of the Physics Department, said: "We are very excited about the potential applications of this multi-disciplinary approach, which harnesses expertise from physics, chemistry, biology, engineering and medicine. We hope it will lead to the development of a new generation of biological imaging and sensing techniques that underpin improvements in healthcare for a range of diseases."
The team also believes FRET microscopy with gold nanoparticles could be used to improve food and water safety. Co-worker Dr Jun Yu, of the Strathclyde Institute of Pharmacy and Biomedical Sciences, said: "This new approach to imaging RNA at a single-cell level may also allow scientists to develop new methods to identify various microbes which may have contaminated food and water. Food safety is a global challenge and using novel nanoprobes to detect food contamination by various microbes will open up a new way of addressing this crucial issue."
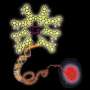
Gold nanoparticles – less than 1000th of the width of a human hair – have a number of advantages over organic dye molecules that are used at present for studying cells with fluorescence microscopy. They are more photostable – meaning they are unchanged by exposure to light – are more sensitive because they can probe over a longer distance, and are less toxic to cells.
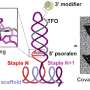
Dr Chen said: "The nanoprobes are based on a type of 'molecular handshake', called Förster resonance energy transfer – or FRET, in which gold nanoparticles are linked with a fluorescent protein, via a hairpin-structured single stranded DNA. Upon interacting with the target mRNA in the cell, the hairpin structure dissolves and a fluorescent signal occurs – enabling the tracking and quantification of the disease-related mRNA at a cellular level, even down to the level of single molecules."
Scientists believe they can be used to deliver other molecules, such as cancer drugs, directly to disease tissues – bypassing normal, healthy cells. Also, they are economical to produce because they only use a tiny trace of the precious metal.
The 18-month project, backed with £119,000 investment from the Biotechnology and Biological Sciences Research Council. It aims to develop a new approach for imaging message ribonucleic acids (mRNA) – a kind of nucleic acid present in all living cells that carries genetic codes from DNA to make protein. By examining key mRNAs at a cellular level, scientists could be able to detect diseases – such as cancer – at an early stage, and to study how effective a particular treatment is.
Provided by University of Strathclyde, Glasgow