March 18, 2013 feature
Please Don't Eat the Daisies: The macroevolution of alternate plant defense strategies
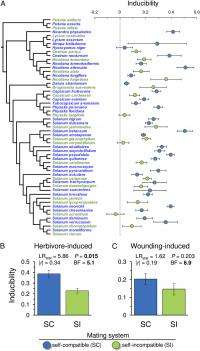
(Phys.org) —As is the case in all areas of science, our understanding of evolutionary biology is… well, evolving. Two such areas are macroevolution (any evolutionary change at or above the level of species – that is, on a scale of separated gene pools) and microevolution (any evolutionary change below the level of species, such as the effect of changes in allele frequency on phenotype). Interestingly, macroevolution and microevolution can be seen as describing fundamentally identical processes on different time scales. Through their divergent effects on population genetics, sexual reproduction strategies could significantly influence phenotypic expression – but the impact of mating system transitions has not been well understood. Recently, however, scientists at Cornell University have shown that the repeated, unidirectional transition from self-incompatibility to self-compatibility (increased inbreeding) leads to the evolution of an inducible (a gene whose expression is responsive to environmental change) – as opposed to a constitutive (a gene that is always expressed) – strategy of plant resistance to herbivores, as well as a strategy in which the loss of self-incompatibility is associated with the evolution of increased specificity in induced plant resistance. Moreover, they demonstrate that these two defense strategies represent evolutionary alternatives, leading to a macroevolutionary tradeoff whose magnitude is dependent on the mating system. The scientists conclude that the evolution of sexual reproductive variation may have profound effects on plant–herbivore interactions, suggesting a new hypothesis for the evolution of two primary plant defense strategies.
Dr. Stuart A. Campbell describes the research that he and Dr. André Kessler conducted and the challenges they encountered. "There were two main challenges inherent in this study," Campbell tells Phys.org. "First, when looking across such a broad swath of evolutionary history – in this case, the evolution of a plant family – we need to know that the mating transitions have occurred repeatedly, and independently. If the transition only happened a few times, we'd be less confident that it was the specifically the mating transition that was the important factor in the evolution of the defense." The researchers were able to sample over 20 such transitions, which Campbell says gave their experiment a lot of power.
"Second, since the defensive chemicals used by different species are extremely different, we need a way to assess the defense that is independent of all the differences among the species – and we wanted to know whether there was any similarity in the way they used those very different chemical arsenals." To get around this, they measured the plants' defenses using herbivorous caterpillars: Using real animals meant that they measured everything that is truly defensive about the plants, regardless of what the defensive chemical actually is, Campbell points out, so caterpillar growth and survival on the plants can thereby be used as a very sensitive measure of all the defensive features of those plants.
"Demonstrating that inducible and constitutive defense strategies represent evolutionary alternatives across many species like this has been notoriously difficult," Campbell continues, "despite the fact that the relationship represents a fundamental assumption in many theories on the evolution of defense. A major reason for this is simply the difficulty of estimating defense using a common metric – it is likely that by using a caterpillar to estimate the true resistance, we had a very sensitive and controlled way of measuring defense that allowed us to detect the relationship."
These challenges required a new perspective. "The key insight in this study was the hypothesis that the mating systems of plants could be such a consistently influential force," Campbell notes. "We had to look beyond the known immediate effects that arise when you have shifts in mating system, and imagine how these immediate changes might alter natural selection on the plants defense." After that, Campbell adds, they relied on the careful implementation of fairly conventional techniques to conduct the experiments.
The research also led to some unexpected results, including the loss of self-incompatibility also being associated with the evolution of increased specificity in induced plant resistance. "This was a rather surprising and incidental part of our study," Campbell explains. "At the beginning, we wanted to look at how plants respond to both real biological damage, but also artificial damage. We realized that having these two measures of plant responses allowed us to ask whether plants had evolved the ability to distinguish these two types of wounding. We compared the two mating systems for this ability and found a difference. "
The question then, says Campbell, was why the self-pollinating plants should exhibit a greater ability to distinguish different damage types. "It may come down to the fact that the mating systems are clearly different in their ability to respond – they're more inducible overall, because it's costly for them to have defense expressed constantly, and so it makes logical sense for them to also avoid mistakes in their responses by evolving mechanisms to discern exactly when a real herbivore is munching on them." In other words, it doesn't make much sense to have a sensitive response to a caterpillar if you can't tell that you're being eaten by one.
Prior theories on why plants should evolve a strategy of being very inducible (as opposed to constitutively defended) have not always been successful in identifying the factors that might systematically dictate when this should happen. "Our study shows how a factor like a reproductive system might create the opportunity for the evolution of non-reproductive plant characteristics," notes Campbell.
"The main importance of this result is its apparent broad applicability," Campbell adds. "Hypotheses for the evolution of plant defense are not usually tested at such a broad scale. In addition, the evolution of inducible defenses is thought to arise from the combination of herbivore frequency and physiological costs, etc; but the exact combination of these that leads to the evolution of inducibility might differ between species. Our hypothesis is interesting in part because it may point toward a more fundamental process (reproductive strategy) that may influence all of these other hypothesized factors."
Regarding next steps in their research, the scientists are currently seeking to understand two aspects of their study's evolutionary results. "First, what is the chemical basis? We've been busy analysing all the species used in this study in order to determine how plants regulate the expression of the chemical arsenal they use in defense against herbivores." For this, the researchers are using modern analytical chemistry techniques, which they will have to couple with new statistical techniques.
"Second, can we capture this co-evolution in action? We've been using field experiments to measure natural selection on both mating and defense, and looking at both pollinators and herbivores, because these are likely to be the animals responsible for imposing that natural selection."
In addition, other areas of research might benefit from this research. "While our study is primarily evolutionary, our results may be useful to other fields. In particular, plant chemists are always interested in knowing what species are likely to have novel compounds. Many plant chemicals have ostensibly evolved to deter herbivores, but may also have interesting biomedical properties – and we think that there is a chemical basis to our results which might inform such studies. From an ecological and conservation standpoint," Campbell concludes, "we know that habitats are becoming increasingly fragmented and disturbed by human activity. This fragmentation may contribute to the evolutionary pattern we see here."
More information: Plant mating system transitions drive the macroevolution of defense strategies, PNAS March 5, 2013 vol. 110 no. 10 3973-3978, doi:10.1073/pnas.1213867110
Journal information: Proceedings of the National Academy of Sciences
Copyright 2013 Phys.org
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of Phys.org.

















