February 15, 2013 feature
We are living in a bacterial world, and it's impacting us more than previously thought
(Phys.org)—Throughout her career, the famous biologist Lynn Margulis (1938-2011) argued that the world of microorganisms has a much larger impact on the entire biosphere—the world of all living things—than scientists typically recognize. Now a team of scientists from universities around the world has collected and compiled the results of hundreds of studies, most from within the past decade, on animal-bacterial interactions, and have shown that Margulis was right. The combined results suggest that the evidence supporting Margulis' view has reached a tipping point, demanding that scientists reexamine some of the fundamental features of life through the lens of the complex, codependent relationships among bacteria and other very different life forms.
The project to review the current research on animal-bacterial interactions began when some scientists recognized the importance of bacteria in their own fields of study. For Michael Hadfield, Professor of Biology at the University of Hawaii at Manoa, the recognition grew over many years while studying the metamorphosis of marine animals. He found that certain bacteria influence marine larvae to settle to particular places on the sea floor, where they transform into juveniles and live out the rest of their lives.
"Once we determined that specific biofilm bacteria provide an essential and unique ligand to stimulate the larvae of one globally distributed marine worm, our research naturally progressed to a study of the portion of the bacterial genome responsible for the signaling, and to other species, where we found the same genes involved," Hadfield told Phys.org. "Coming from different perspectives on the study of animal-bacterial interactions, and recognizing many more, Margaret McFall-Ngai [Professor of Medical Microbiology and Immunology at the University of Wisconsin, Madison] and I discussed the current situation extensively and then decided to attempt to draw together a significant number of experts on various approaches to the study of bacterial-animal interactions to draft a paper such as the one you have in hand. We proposed a 'catalysis meeting' on the subject to the National Science Foundation's National Evolutionary Synthesis Center (NESCent), which was funded, and the project took off."
Bacteria surround us
In many respects, it's easy to see the prominent role that bacteria play in the world. Bacteria were one of the first life forms to appear on Earth, about 3.8 billion years ago, and they will most likely survive long after humans are gone. In the current tree of life, they occupy one of the three main branches (the other two are Archaea and Eucarya, with animals belonging to the latter). Although bacteria are extremely diverse and live nearly everywhere on Earth, from the bottom of the ocean to the inside of our intestines, they have a few things in common. They are similar in size (a few micrometers), they are usually made of either a single cell or a few cells, and their cells don't have nuclei.
Although scientists have known for many years that animals serve as a host for bacteria, which live especially in the gut/intestines, in the mouth, and on the skin, recent research has uncovered just how numerous these microbes are. Studies have shown that humans have about 10 times more bacterial cells in our bodies than we have human cells. (However, the total bacteria weigh less than half a pound because bacterial cells are much smaller than human cells.)
While some of these bacteria simply live side-by-side with animals, not interacting much, some of them interact a lot. We often associate bacteria with disease-causing "germs" or pathogens, and bacteria are responsible for many diseases, such as tuberculosis, bubonic plague, and MRSA infections. But bacteria do many good things, too, and the recent research underlines the fact that animal life would not be the same without them.
"The true number of bacterial species in the world is staggeringly huge, including bacteria now found circling the Earth in the most upper layers of our atmosphere and in the rocks deep below the sea floor," Hadfield said. "Then add all of those from all of the possible environments you can think of, from cesspools to hot springs, and all over on and in virtually every living organism. Therefore, the proportion of all bacterial species that is pathogenic to plants and animals is surely small. I suspect that the proportion that is beneficial/necessary to plants and animals is likewise small relative to the total number of bacteria present in the universe, and surely most bacteria, in this perspective, are 'neutral.' However, I am also convinced that the number of beneficial microbes, even very necessary microbes, is much, much greater than the number of pathogens."
Animal origins and coevolution
From our humble beginnings, bacteria may have played an important role by assisting in the origins of multicellular organisms (about 1-2 billion years ago) and in the origins of animals (about 700 million years ago). Researchers have recently discovered that one of the closest living relatives of multicellular animals, a single-celled choanoflagellate, responds to signals from one of its prey bacterium. These signals cause dividing choanoflagellate cells to retain connections, leading to the formation of well-coordinated colonies that may have become multicellular organisms. However, such questions of origin have been subjects of intense debate, and scientists have many hypotheses about how these life forms emerged. A bacterial role in these processes does not exclude other perspectives but adds an additional consideration.
After helping get animals started, bacteria also played an important role in helping them along their evolutionary path. While animal development is traditionally thought to be directed primarily by the animal's own genome in response to environmental factors, recent research has shown that animal development may be better thought of as an orchestration among the animal, the environment, and the coevolution of numerous microbial species. One example of this coevolution may have occurred when mammals evolved endothermy, or the ability to maintain a constant temperature of approximately 40 °C (100 °F) by metabolic means. This is also the temperature at which mammals' bacterial partners work at optimum efficiency, providing energy for the mammals and reducing their food requirement. This finding suggests that bacteria's preferred temperature may have placed a selection pressure on the evolution of genes associated with endothermy.
Bacterial signaling
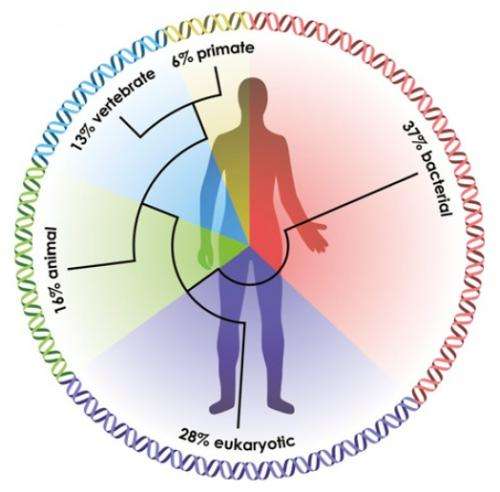
Evidence for a deep-rooted alliance between animals and bacteria also emerges in both groups' genomes. Researchers estimate that about 37% of the 23,000 human genes have homologs with bacteria and Archaea, i.e., they are related to genes found in bacteria and Archaea that were derived from a common ancestor.
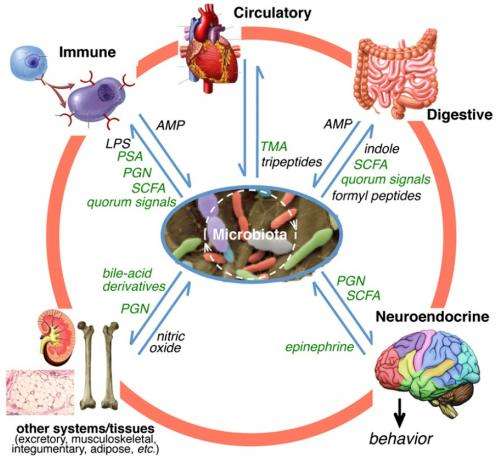
Many of these homologous genes enable signaling between animals and bacteria, which suggests that they have been able to communicate and influence each other's development. One example is Hadfield and his group's discovery that bacterial signaling plays an essential role in inducing metamorphosis in some marine invertebrate larvae, where the bacteria produce cues associated with particular environmental factors. Other studies have found that bacterial signaling influences normal brain development in mammals, affects reproductive behavior in both vertebrates and invertebrates, and activates the immune system in tsetse flies. The olfactory chemicals that attract some animals (including humans) to their prospective mates are also produced by the animals' resident bacteria.
Bacterial signaling is not only essential for development, it also helps animals maintain homeostasis, keeping us healthy and happy. As research has shown, bacteria in the gut can communicate with the brain through the central nervous system. Studies have found that mice without certain bacteria have defects in brain regions that control anxiety and depression-like behavior. Bacterial signaling also plays an essential role in guarding an animal's immune system. Disturbing these bacterial signaling pathways can lead to diseases such as diabetes, inflammatory bowel disease, and infections. Studies also suggest that many of the pathogens that cause disease in animals have "hijacked" these bacterial communication channels that originally evolved to maintain a balance between the animal and hundreds of beneficial bacterial species.
Signaling also appears in the larger arena of ecosystems. For example, bacteria in flower nectar can change the chemical properties of the nectar, influencing the way pollinators interact with plants. Human infants who are born vaginally have different gut bacteria than those delivered by Caesarean section, which may have long-lasting effects. And bacteria feeding on dead animals can repel animal scavengers—organisms 10,000 times their size—by producing noxious odors that signal the scavengers to stay away.
In the gut
In the earliest animals, gut bacteria played an important role in nutrition by helping animals digest their food, and may have influenced the development of other nearby organ systems, such as the respiratory and urogenital systems. Likewise, animal evolution likely drove the evolution of the bacteria, sometimes into highly specialized niches. For example, 90% of the bacterial species in termite guts are not found anywhere else. Such specialization also means that the extinction of every animal species results in the extinction of an unknown number of bacterial lineages that have evolved along with it.
Scientists have also discovered that bacteria in the human gut adapts to changing diets. For example, most Americans have a gut microbiome that is optimized for digesting a high-fat, high-protein diet, while people in rural Amazonas, Venezuela, have gut microbes better suited for breaking down complex carbohydrates. Some people in Japan even have a gut bacterium that can digest seaweed. Researchers think the gut microbiome adapts in two ways: by adding or removing certain bacteria species, and by transferring the desired genes from one bacterium to another through horizontal gene transfer. Both host and bacteria benefit from this kind of symbiotic relationship, which researchers think is much more widespread than previously thought.
The big picture
Altogether, the recent studies have shown that animals and bacteria have histories that are deeply intertwined, and depend on each other for their own health and well-being as well as that of their environments. Although the researchers focused exclusively on animal-bacteria interactions, they expect that similar trends of codependency and symbiosis are universal among and between other groups, such as Archaea, fungi, plants, and animals. Once considered an exception, such intermingling is now becoming recognized as the rule—just as Margulis predicted many decades ago. Due to these symbiotic relationships, the scientists here propose that the very definitions of an organism, an environment, a population, and a genome have become blurred and should be reviewed. It may be, for instance, that animals are better viewed as host-microbe ecosystems than as individuals.

In addition, the scientists predict that the recent findings on animal-bacteria interactions will likely require biologists to significantly alter their view of the fundamental nature of the entire biosphere. Along these lines, large-scale research projects such as the Human Microbiome Project and the Earth Microbiome Project are already underway to investigate the wide range of bacteria in the individual and global systems, and to see what happens when the bacteria are disturbed.
In the end, the scientists hope that the results will promote more cross-disciplinary collaboration among scientists and engineers from different fields to explore the new microbial frontier. They argue that these discoveries should revolutionize the way that biology is taught from the high school level on up, by focusing more on the relationships between bacteria, their animal partners, and all other life forms.
"It is hard to summarize a single 'most important conclusion,' other than the admonition to biologists studying animals, from behavior to physiology and ecology to molecular biology, that no matter what process you think you are studying, you must look for and consider a major role for bacteria," Hadfield said. "In many cases, this may require partnerships across traditional boundaries of research, meaning that zoologists must collaborate with microbiologists to advance their research, that molecular biologists must collaborate with whole-organism biologists, etc. We want badly for the message in 'Animals in a bacterial world,' to be a call for the necessary disappearance of the old boundaries between life science departments (e.g., Depts of Zoology, Botany, Microbiology, etc.) in universities, and societies (e.g., the American Society for Microbiology, etc.). We also want the message disseminated in college and university classes from introductory biology to advanced courses in the various topic areas of our paper."
The results will profoundly change the way that the scientists of this collaboration continue with their own areas of research, Hadfield said.
"Each of the authors of our paper conducts basic research in one or more areas of animal-bacterial interactions discussed in the paper, and each will continue to focus on her/his own speciality, I'm sure," he said. "However, I'm also certain that the interactions developed during the composition and writing of the paper (starting with our NESCent meeting in October 2011, when most of us met for the first time) will impact our own research and cause us to establish new collaborations with other laboratories. That has already occurred for me; I have a new collaboration with Dianne Newman's group at CalTech, an outstanding group of bacteriologists who are helping us do a much more in-depth investigation of the bacterial gene-products responsible for larval development."
More information: Margaret McFall-Ngai, et al. "Animals in a bacterial world, a new imperative for the life sciences." PNAS Early Edition. DOI: 10.1073/pnas.1218525110
Journal information: Proceedings of the National Academy of Sciences
Copyright 2013 Phys.org
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of Phys.org.