Highly efficient production of advanced biofuel by metabolically engineered microorganism
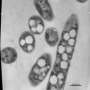
A Korean research team led by Distinguished Professor Sang Yup Lee at the Department of Chemical and Biomolecular Engineering, KAIST, applied a systems metabolic engineering approach to improve the production of butanol through enhancing the performance of Clostridium acetobutylicum, one of the best known butanol-producing bacteria.
Fuels including gasoline, diesel, and jet fuel are derived from fossil oil thorough the petroleum refinery processes. Increased concerns over environmental problems and limited fossil resources drive scientists and researchers to turn their attention to developing fossil-free, bio-based processes for the production of fuels from renewable non-food biomass. Utilizing systems metabolic engineering, a Korean research team at the Korea Advanced Institute of Science and Technology (KAIST) has succeeded in demonstrating an optimized process to increase butanol production by generating an engineered bacterium.
In the paper published in mBio, a broad-scope, online-only, and open access journal issued by the American Society for Microbiology (ASM), on October 23 as a featured article, Distinguished Professor Sang Yup Lee at the Department of Chemical and Biomolecular Engineering, KAIST, a premier science and engineering university in Korea, Dr. Do Young Seung at GS Caltex, a large oil refining company in Korea, and Dr. Yu-Sin Jang at BioFuelChem, a startup butanol company in Korea, applied a systems metabolic engineering approach to improve the production of butanol through enhancing the performance of Clostridium acetobutylicum, one of the best known butanol-producing bacteria.
Microorganisms have proven to be efficient biocatalysts for the production of biofuels from various sources of biomass in an environmentally-friendly way. However, the microorganisms isolated from nature are often inefficient for the economical production of desired products at an industrial scale. Thus microorganisms' performance needs to be improved in order to be suitable for the industrial production of biofuels. Systems metabolic engineering, which allows metabolic engineering at a systems-level, is employed for designing and optimizing cellular metabolic and regulatory networks to induce the most efficient production of target bioproducts.
Butanol has been used as an important industrial solvent, and is also a great alternative fuel because of its similar properties to gasoline. It can also be blended with gasoline at any ratio. Butanol is naturally produced by some anaerobic bacteria, but the efficiency of its production could not match that of ethanol due to the high toxicity of butanol to host organisms and the production of byproducts such as acetone and organic acids. Over the past decades, many research groups extensively studied clostridial butanol producing organisms to achieve higher yield and titer, but the limited genetic modification tools and complex metabolic pathways of clostridia hampered the successful development of an engineered strain capable of producing butanol at a higher yield and titer.
The Korean research team analyzed metabolic pathways leading to butanol production and found that two different solvent-forming pathways can be potentially employed. In one pathway, butanol is directly produced from carbon source, which was termed as hot channel, and in the other, butanol is converted from the acids produced earlier in fermentation process, which was termed as cold channel. Using the in silico modeling and simulation tools, Professor Lee's team demonstrated that the hot channel allowed a much better approach to produce butanol compared with the cold channel. To reinforce a metabolic flux toward the hot channel for butanol production, the metabolic network of C. acetobutylicum strain was systematically engineered.
In addition, the downstream process was optimized and an in situ recovery process was integrated to achieve higher butanol titer, yield, and productivity. The combination of systems metabolic engineering and bioprocess optimization resulted in the development of a process capable of producing more than 585 g of butanol from 1.8 Kg of glucose, which allows the production of this important industrial solvent and advanced biofuel to be cost competitive.
More information: The paper, "Enhanced butanol production obtained by reinforcing the direct butanol-forming route in Clostridium acetobutylicum, can be viewed at dx.doi.org/10.1128/mBio.00314-12
Journal information: mBio