January 27, 2012 report
New study shows prions able to jump between species more easily than thought
(PhysOrg.com) -- A group of French researchers has found that prions are more easily able to jump between species than has been previously thought. In their paper published in Science, they show that prions from other species, implanted in the brains of mice showed up in other organs after a period of time, suggesting that brain autopsies are not sufficient to detect the presence of the disease. British neurology researcher John Collinge has also published a perspective on the topic in the same journal.
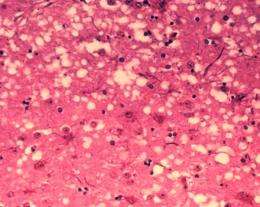
Prions are infectious pathogens that are made up mainly of a form of misfolded PrP proteins. Infections in different species go by different names, the most famous being “mad cow disease” in cows. In humans, it’s called Creutzfeldt-Jakob disease and results in clumps of brain tissue dying resulting in dementia and other symptoms and eventually death.
Up until now, it has been thought that there existed a biological barrier that made it difficult for prions to pass from one species of animal to another, but that assumption has been based mainly on brain autopsies. In this new study, the researchers found that when prions from other species (elk, hamsters and cattle) were inserted into the brains of mice that had been genetically altered to express the human or sheep version of PrP, the transfer rate to the brain was very small (just 3 out of 43 cases) as expected. However, when they also autopsied other organs in the mice such as tonsils and especially the spleen they found that the prions had migrated in 26 of 41 cases. They also found that in such cases, the mice didn’t display any symptoms of the disease which leads the authors to conclude that many more animals and humans likely carry the disease than has been thought.
The concern is that human carriers could be inadvertently infecting others through blood transfusions, organ donation or even via surgical instruments since prions have been found to be resistant to normal antibacterial processes. And if that is the case, it appears likely that at least some of those infected would eventually find themselves falling prey to Creutzfeldt-Jakob disease.
These new findings come on the heels of recent news that Britain’s NHS National Prion Clinic has sent out messages to neurologists across the country indicating that a new blood test has become available, which has come none too soon as a recent survey of tissue samples taken from removed appendixes showed that as many as 1 in 4,000 people in the UK could be carriers.
More information: Facilitated Cross-Species Transmission of Prions in Extraneural Tissue, Science 27 January 2012: Vol. 335 no. 6067 pp. 472-475. DOI:10.1126/science.1215659
ABSTRACT
Prions are infectious pathogens essentially composed of PrPSc, an abnormally folded form of the host-encoded prion protein PrPC. Constrained steric interactions between PrPSc and PrPC are thought to provide prions with species specificity and to control cross-species transmission into other host populations, including humans. We compared the ability of brain and lymphoid tissues from ovine and human PrP transgenic mice to replicate foreign, inefficiently transmitted prions. Lymphoid tissue was consistently more permissive than the brain to prions such as those causing chronic wasting disease and bovine spongiform encephalopathy. Furthermore, when the transmission barrier was overcome through strain shifting in the brain, a distinct agent propagated in the spleen, which retained the ability to infect the original host. Thus, prion cross-species transmission efficacy can exhibit a marked tissue dependence.
PERSPECTIVE: The Risk of Prion Zoonoses, Science 27 January 2012: Vol. 335 no. 6067 pp. 411-413. DOI:10.1126/science.1218167
Journal information: Science
© 2011 PhysOrg.com