December 7, 2010 feature
Researchers improve efficiency of low-cost solar cells
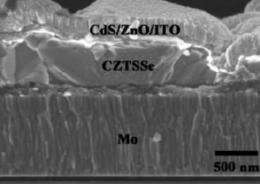
(PhysOrg.com) -- As part of the recent progress in improving solar cells for widespread use, researchers from Purdue University have designed solar cells made of low-cost, abundant materials that are easily scalable and very stable. The researchers have increased the solar cells' total area efficiency to 7.2% and plan to make further improvements in the future.
The researchers, Qijie Guo, Grayson M. Ford, Wei-Chang Yang, Bryce C. Walker, Eric A. Stach, Hugh W. Hillhouse, and Rakesh Agrawal, have published their study on the improved solar cells in a recent issue of the Journal of the American Chemical Society. They fabricated the solar cells from copper-zinc-tin-chalcogenide (CZTSSe), which is an Earth-abundant material, using a solution-based thin-film deposition method. Previous research has shown that these methods can provide high yields at lower manufacturing costs compared to other methods.
The solar cell design is based on the researchers' earlier study in which they demonstrated that solar cells fabricated using CZTS nanocrystals are potentially viable, although they had efficiencies of less than 1%. Here, the researchers made significant improvements to the design by tuning the composition of the nanocrystals as well as developing a more robust thin-film coating method.
After synthesizing the nanocrystals and applying them on a substrate for a total film thickness of 1 micrometer, the researchers observed that the nanocrystal film featured large, densely packed grains, which leads to improved solar cell efficiency. In testing, the solar cells could achieve a total area efficiency of 7.2%. As coauthor Hugh Hillhouse explained, the total area efficiency refers to the entire cell, rather than just the “active area.”
"It is the total area efficiency that matters most," he told PhysOrg.com. “Some people report an ‘active area’ efficiency, which only includes areas that the light reaches. However, all thin film solar cells are made with metal contacts that block the light from reaching some areas. When you include this loss, we use the term ‘total area’ efficiency. It is the most fair and important efficiency.”
The 7.2% efficiency was reached after “light soaking” for 15 minutes under one-sun illumination; when the light was turned off, the efficiency dropped to 6.89%.
“Light soaking simply means that we shine normal intensity simulated sunlight on the cell for a period of time before we make the measurement,” Hillhouse said. “Most likely, the light soaking allows photogenerated carriers to fill traps, shift the quasi-Fermi levels, and/or screen barriers created by band offsets. It doesn’t present a problem since real solar cells are naturally light soaked – they sit in the sun.”
Although currently there are no CZTS or CZTSSe solar cells on the market for comparison, the solar cells in this study are very competitive with other fabrication methods.
“The best cells formed by vacuum processes have only reached 6.7%,” Hillhouse said. “Typically, solar cells produced by vacuum-based processes have been more efficient, but also more expensive. For the case of CZTS, the solution phase approach (our nanocrystal route and IBM’s hydrazine route) is more efficient.”
One potential area for improvement for these solar cells lies in improving their low quantum efficiency for light of longer wavelengths (i.e., the near-infrared range). The researchers attempted to improve this efficiency by increasing the thickness of the absorber, although their initial experiments showed that thicker absorber layers also had increased resistance. In the future, they plan to optimize the fabrication for thicker films, which could further increase the overall efficiency.
“There is a lot of compositional freedom in the CZTSSe system, and it is likely that the optimum compositions, device structure, and processing conditions have not yet been found – but we are working on it,” Hillhouse said.
More information: Qijie Guo, et al. “Fabrication of 7.2% Efficient CZTSSe Solar Cells Using CZTS Nanocrystals.” J. Am. Chem. Soc. ASAP. DOI: 10.1021/ja108427b
Copyright 2010 PhysOrg.com.
All rights reserved. This material may not be published, broadcast, rewritten or redistributed in whole or part without the express written permission of PhysOrg.com.



















