Watching nanosheets and molecules transform under pressure could lead to stronger materials
(PhysOrg.com) -- When it comes to tests of strength, graphite -- actually layered sheets of carbon atoms -- fares badly. Subject it to ultra-high pressure, though, and graphite becomes diamond, the hardest substance known, and a uniquely useful material in a variety of applications.
But while diamonds may be forever, most materials that transform under high pressure revert to their original structure when the pressure is lifted -- losing any useful properties they may have gained when the squeeze was on.
Now, by understanding the process behind the transformation itself, from both experimental and theoretical perspectives, researchers have taken a potential step toward creating a new class of exceptionally strong, durable materials that maintain their high-pressure properties -- including strength and superconductivity -- in everyday low-pressure environments.
The research, led by Zhongwu Wang, staff scientist at the Cornell High Energy Synchrotron Source (CHESS) and including Roald Hoffmann, the 1981 chemistry Nobel laureate and Frank H.T. Rhodes Professor of Humane Letters Emeritus, appears in the Oct. 12, issue of the Proceedings of the National Academy of Sciences.
Additional scientists at CHESS, a group in Korea and a postdoctoral associate in the Hoffmann group, Xiao-dong Wen, also contributed.
Researchers frequently use X-ray diffraction, a technique in which X-rays are projected at a structure and captured on film after they pass through or bounce off its surfaces, to determine the static structures of atoms and molecules. But until now, the transformation and interaction between two structures happened in a metaphorical black box, said Wang.
To open the box, researchers focused on wurtzite, a cadmium-selenium crystal in which atoms are arranged in a diamondlike structure and molecules are bonded on the surface. When thin sheets of wurtzite are squeezed under 10.7 gigapascals of pressure, or 107,000 times the pressure on the Earth's surface, their atomic structure transforms into a rock salt-like structure
Subjecting a macro-sized crystal to high pressure can cause it to break (small defects in the crystal structure magnify, causing the structure, and the transformation process, to become irregular) -- so the group's Korean collaborators instead prepared wurtzite nanosheets, which are just 1.4 nanometers thick and defect-free.
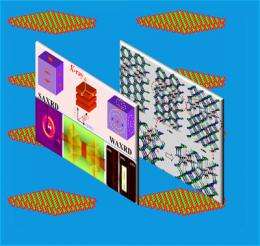
As pressure was applied, Wang and colleagues integrated two X-ray diffraction techniques (small- and large-angle X-ray diffraction) to characterize changes in the crystal's surface shape and interior atomic structure, as well as the structural change of surface-bonded molecules.
They first discovered that the nanosheets required three times the pressure to undergo the transformation as the same material in larger crystal form.
They also tested the material's yield strength (the stress level at which it begins to deform), hardness (resistance to scratching or abrading) and elasticity (ability to return to its original form) during the transformation. Understanding how those properties change as the molecules interact could help researchers design stronger, tougher materials, Wang said.
And adding a bonding molecule called a soft ligand to the surface of the high-pressure nanosheets, the researchers observed the effect of that bonding to the nanosheets' internal structure, transformation pressure, and spacing.
Meanwhile, as Wang and colleagues performed the experiments at CHESS, Wen and Hoffmann worked on the corresponding theory behind the transformation interaction.
"Both the experiment and the simulation agree well," Wang said. "Now we know how the atoms move. We understand the intermediate procedure."
The next step is to test ways of blocking the reverse transformation from rock salt back to wurtzite, creating a material that maintains rock salt's unique properties under ambient pressure.
And Wang's experimental process could hold promise for understanding the transformation pathway for other compounds as well.
"It can apply to all other materials," Wang said. "Just follow our way of measurement."
Provided by Cornell University