Newly Discovered Fat Molecule: An Undersea Killer with an Upside
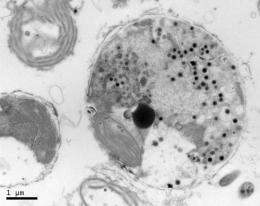
(PhysOrg.com) -- A chemical culprit responsible for the rapid, mysterious death of phytoplankton in the North Atlantic Ocean has been found by collaborating scientists at Rutgers University and the Woods Hole Oceanographic Institution (WHOI). This same chemical may hold unexpected promise in cancer research.
The team discovered a previously unknown lipid, or fatty compound, in a virus that has been attacking and killing Emiliania huxleyi, a phytoplankton that plays a major role in the global carbon cycle
“Emiliania huxleyi is the rock star of phytoplankton,” explains Kay Bidle, Rutgers assistant professor of marine science in the Institute of Marine and Coastal Sciences. “It blooms all over the oceans, and we can easily see it by satellite. We know that these blooms are frequently infected with viruses, and this virus is specific to this phytoplankton.”
“The lipids are the key ingredient in the virus that causes the phytoplankton to die,” says WHOI scientist Benjamin Van Mooy. “We have a completely different lipid molecule that, as far as we know, is unknown to science.”
E. huxleyi grows rapidly in the North Atlantic, “in these big blooms that you can actually see from outer space,” Van Mooy says.
“But,” adds Van Mooy, “they die just almost as quickly as they start out, and we’re not sure why. They die after a few days.”
Bidle and Assaf Vardi, a postdoctoral investigator in his laboratory and the study’s lead author, had been examining the interaction between the virus and the dying phytoplankton and had developed ideas for how this process works. After Vardi heard lipid expert Van Mooy give a talk in Santa Fe, N.M., he suggested the collaboration between WHOI and Rutgers.
“I saw Ben's talk on marine microbes and lipids…[and] I ran after him,” said Vardi. “We told him about our ideas” involving the virus’s effect on the phytoplankton.
“They studied the viruses and I study lipids,” Van Mooy said. “It seemed like a good mix.”
Their paper is published in the Nov. 6 issue of Science.
E. huxleyi performs photosynthesis—“just like plants,” says Van Mooy. “They suck up carbon dioxide.” In doing so, they reduce the amount of CO2 released into the atmosphere. They form a calcium carbonate shell, also helping to regulate the carbon cycle.
If viruses are killing off phytoplankton, this can increase greenhouse emissions, Van Mooy suggests. “That’s important because if viruses infect a whole bunch of cells, then they can’t perform photosynthesis, they can’t take up carbon dioxide.”
In April 2008, Van Mooy’s team visited the sites of E. huxleyi blooms during a research cruise between Woods Hole and Bermuda and collected samples for lipid analysis back in the laboratory.
They immediately recognized lipids that were just like those in virally infected E. huxleyi cells grown by the Rutgers team. Helen Fredricks, a research associate with Van Mooy, carried out the lipid analyses at WHOI. “Seeing this viral lipid appear during the course of infection was amazing, and then we found it in the ocean too. We were celebrating in the lab that day.”
Adds Vardi: "Viruses are really important players in regulating phytoplankton blooms. We zoom into the bloom and try to understand the interaction between the viruses and host, which is this really important, cosmopolitan, bloom-forming species.”
After isolating the viral lipids, the team found that the lipids alone were able to bring about the symptoms of viral infection in the phytoplankton. “The lipids themselves act just like the virus,” says Van Mooy. “We can cause the phytoplankton to die by just giving the lipids.”
This alone was enough to excite the team. “Now we have a biological marker that we can go out on a ship and look for and identify where this [infection of phytoplankton] is happening and learn how to study it better,” Van Mooy says.
But there may be other, even farther-reaching implications. Both the virus and the newly found lipid deal their deadly blow by causing the upper-ocean plants to commit cellular suicide. As a major focus of their research at Rutgers, Bidle’s lab has found that “programmed cell death” is an important process in the fate of marine phytoplankton and in the demise of blooms in the oceans. Bidle’s group had previously found that successful infection of E. huxleyi induced, and actually required, the programmed cell death pathway.
But programmed cell death is not unique to phytoplankton. It is a common and healthy process in all kinds of cells, including human cells.
According to Vardi, “These lipids can induce programmed cell death in many organisms, including animals and plants. They also enrich in plasma membrane, and they are the port of the cell, where pathogens get in and out of the cell. This is important in viral diseases.”
There is also a potential connection with cancer. If a healthy cell is stressed or damaged, usually it will kill itself with programmed cell death. But cancer cells have a defect: “They don’t kill themselves,” says Bidle.
“It’s a critical aspect of cancer research, because cancer cells have figured out a way to turn off the programmed cell death pathway,” he says. “In cancer studies, they try to figure out ways to reactivate those pathways.”
The lipid may help shed light on why cancer cells are unable to commit suicide. Someday, the researchers say, it might suggest ways to correct that defect. Right now, the lipid is only known to be effective in algae, but in the future, the team is hoping to test the effectiveness of their molecule in experiments with cancer cells.
More immediately, scientists hope to learn more about the central role phytoplankton—and viruses—play in regulating climate. Bidle says this is a particularly interesting virus. “It appears that the virus has…borrowed, copied actually, the genes for this lipid from the host,” he says. “Similar genes are still on the host, but the virus has figured out a way to take those genes and put them into its own genome, and alter them enough to make them more toxic."
”We find the biosynthetic pathway for this unique lipid encoded in the virus genome, not only in the host, and this has never been described before in any other virus,” Vardi says. “We knew that [lipids] were important, but we were really intrigued about why the virus contained these genes. And what is the role of the pathway in the co-evolution of programmed cell death in the host and virus.”
Van Mooy sees it as a struggle between two mighty forces. “The phytoplankton are at one end of the boxing ring and they’re taking up carbon dioxide, and the viruses are at the other end, and they’re out to kill them. And how that works out controls how much carbon dioxide is taken up.
“We’re very interested in understanding what controls these phytoplankton,” he says. “I didn’t know that much about viruses until I started working on this project and the Rutgers researchers didn’t know that much about lipids. So now we’re both really onto something here. We’re continuing to collaborate. “We have found other interesting lipids from these viruses,” says Van Mooy.
“There are probably more out there. And who knows what kind of activities they may be involved with. They may hold a cure for a human disease or they may play unknown role in…phytoplankton.
“I’d like to think [the work] is going to have a continued impact.”
Provided by Woods Hole Oceanographic Institution
















