This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
A new type of metallacrown ether based on polyoxometalate opens research opportunities
Crown ethers were discovered in 1967. They were then modified by adding a metal-containing unit, creating metallacrown ethers. These metallacrown ethers have been the subject of intensive research. Depending on the molecular makeup of the metallacrown ethers and their resultant architecture, the properties and, therefore, the uses of the metallacrowns can change. They have many different uses currently, and ongoing studies continue to expand their application.
Just a few of these include magnetic refrigeration, imaging agents—specifically as potential contrast agents in magnetic resonance imaging—and single-molecular magnetism, which is being explored for its use in quantum computing.
More specifically, "metallacrown ethers have recently attracted considerable attention because they selectively capture guest species, especially metal ions and organic ammonium cations. And this characteristic may increase its utility in catalysis, magnetism, and proton conductors," said Dr. Xiao, the paper's lead author and a scientific researcher at the Department of Chemistry at Tsinghua University, Beijing.
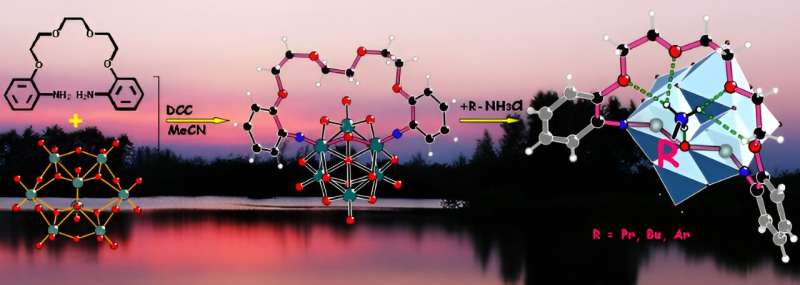
Knowing what can be done with the metallacrown ethers, there is a drive to continue to discover more types. Dr. Xiao, a member of Prof. Wei's group at Tsinghua University in Beijing, has been studying modifying crown ethers using polyoxometalates (POM) as replacing units to create a new type of metallacrown ether.
"Combining crown ether molecules with polyoxometalates would open up broad fields for studies on supermolecular compounds and hybrid materials," said Xiao. And they were successful. "Through covalent modification, polyoxometalates and functional organic compounds were cleverly combined together to achieve further expansion of their functions."
Their research was published in Polyoxometalates.
Crown ethers are ring-shaped molecules containing several ether groups. Their name arises from the ring shape that gives the molecules the look of a crown. On their own, they are strong binding agents for positively charged alkali metal ions.
Metallacrown ethers are a kind of metallamacrocycle complex replacing the ethylene groups of crown ethers partially or wholly with metal-containing units. Compared to crown ether, they may have additional functions in recognition, catalysis, and magnetism.
In this experiment, Dr. Xiao and her partners combined crown ether molecules with polyoxometalates (POM) made up of early transition metal elements by covalent bonds to create a novel metallacrown ether, expanding its uses significantly. The difficulty in this experiment was that they were only partially replacing the ethylene groups, which has been reported before, but it required a different method than the more common full replacement.
As well, they were incorporating POM, which has not been used to create a metallacrown ether before. So, the challenge remained to build a polyoxometalatocrown ether with the ability to capture specific guest molecules.
The host, a larger molecule (in this case, the metallacrown ether), would hopefully capture the guest, a smaller molecule (in this case, ammonia cations), into a non-covalent bond similar to the bonding that occurs in many biological systems such as proteins.
There was some previous evidence that ammonium cations, which are positively charged ammonium ions, and their organic derivatives showed promise as potential guest molecules. "Of course, it is not we who choose the amino cation as the guest, but this kind of metallacrown ether itself. Through some experiments, we got these host-guest compounds," said Xiao.
In essence, they successfully built a polyoxometalatocrown ether that can capture ammonium cations and gained a series of host-guest complexes.
When asked about future research, Xiao said, "The selection of amino cations as guest molecules by metallacrown ether has laid a foundation for us to further research on the function of these compounds, such as recognition, ion exchange, and catalysts." Their next step "is to try to bind functional amino cations: such as all kinds of amino acids and study their related catalytic properties from POM. It is hoped that a good ion recognition agent or a specific catalyst can be screened."
More information: Fengping Xiao et al, Polyoxometalatocrown ether: A new type of metallacrown ether based on polyoxometalate, Polyoxometalates (2024). DOI: 10.26599/POM.2024.9140055
Provided by Tsinghua University Press