This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
trusted source
proofread
Research team introduces new non-toxic method for producing high-quality graphene oxide
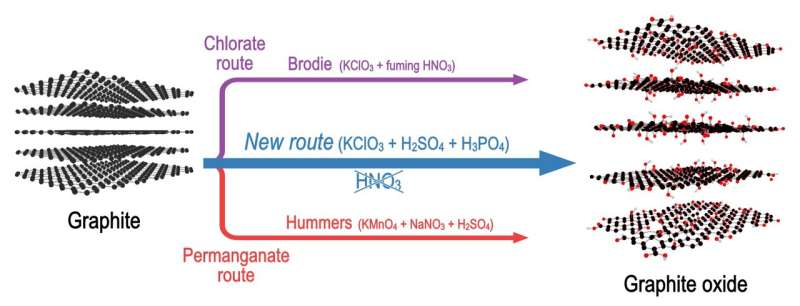
Researchers from Umeå University in Sweden have found a new way to synthesize graphene oxide, which has significantly fewer defects compared to materials produced by the most common method. Similarly good graphene oxide could be synthesized previously only by using a rather dangerous method involving extremely toxic fuming nitric acid.
Graphene oxide is often used to produce graphene by removing oxygen. However, if there are holes in graphene oxide, there will also be holes after it is converted to graphene. Therefore, the quality of the graphene oxide is very important.
Alexandr Talyzin and his research group have now cracked the puzzle of how to safely make good graphene oxide. Their results are published in the journal Carbon.
Graphene is often described as a wonder material thanks to its flexibility, high mechanical strength and conductivity. But all properties of graphene are affected by defects. Graphene produced from graphene oxide has much worse-than-expected mechanical properties and conductivity.
Many studies have demonstrated that synthesis by the most commonly used method, the Hummers method, always results in a significant number of defects. The much older Brodie method provides nearly completely hole-free graphene oxide, but this type of graphene oxide is still not produced by any companies and not available commercially. "It is simply too dangerous and not suitable for industrial manufacturing," says Alexandr Talyzin.
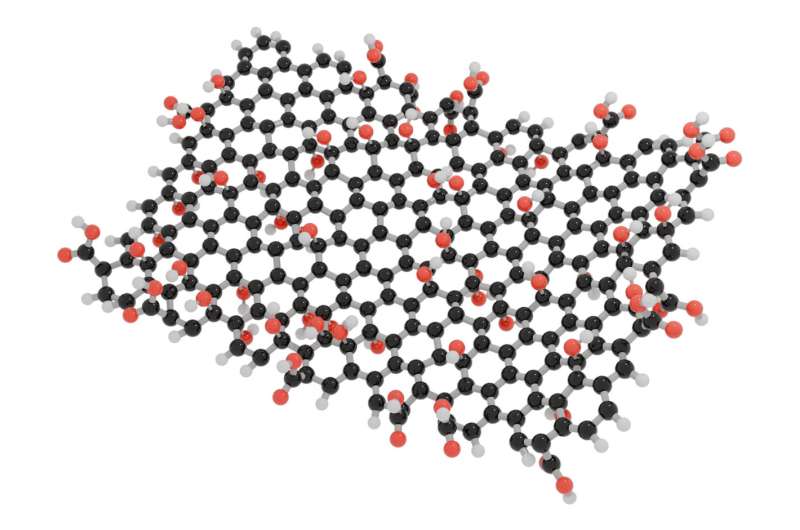
Now, the researchers have found a new method that combines the acid from the Hummers method (H2SO4) and the oxidant from the Brodie method (potassium chlorate), allowing them to produce graphene oxide with a number of defects as small as those in Brodie, but using a synthesis procedure as simple as Hummers oxidation.
"This method should be named after Bartosz Gurzeda, a researcher working in my group... as the Gurzeda method," says Alexandr Talyzin.
According to Talyzin, there are many reasons to believe that the Gurzeda method will become as popular as the Hummers method whenever defect-free graphene oxide is needed. This method is for making graphene by removing oxygen groups or for the preparation of gas protection coatings, semi-permeable membranes, sensors, and many other possible applications.
In the last decade, much interest has also arisen for applications of graphene oxide itself. Layered graphene oxide materials are intensively studied for membrane applications with the dream of producing drinkable water by simple filtration of salts from sea water or creating semi-permeable protective coatings that allow water to pass while keeping away dangerous organic pollutants, such as toluene.
"We want the research society to try and test this new graphene oxide in their applications and see the difference. Graphene oxide is not one material; it is a family of materials with rather different properties, providing us with infinite opportunities for new applications," says Talyzin.
More information: Bartosz Gurzęda et al, Graphite oxide by "chlorate route" oxidation without HNO3: Does acid matter?, Carbon (2024). DOI: 10.1016/j.carbon.2024.118899
Provided by Umea University





















