This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Carbon nanotube membrane unleashes the power of permanganate for superior micropollutant removal
With the rapid development of industrialization, water pollution is becoming more and more serious. The traditional water treatment method can't effectively remove organic pollutants, so advanced oxidation technology has become a possible solution.
As a potential chemical oxidant, permanganate (KMnO4) has been widely studied for water decontamination due to its high efficiency, cost-effectiveness and high stability. However, the poor stability and limited oxidation potential (1.68V) of KMnO4 restrict its applications.
To overcome these problems, researchers have tried various innovative approaches to boost the reactivity of KMnO4. Unfortunately, because of the addition of toxic and expensive chemicals and the occurrence of secondary pollution, these routes strongly impede the scientific progress of KMnO4 oxidation toward practical applications. In recent years, metal-free carbon materials, especially carbon nanotubes (CNT), have emerged as an attractive additive to KMnO4 oxidation due to their environmental friendliness.
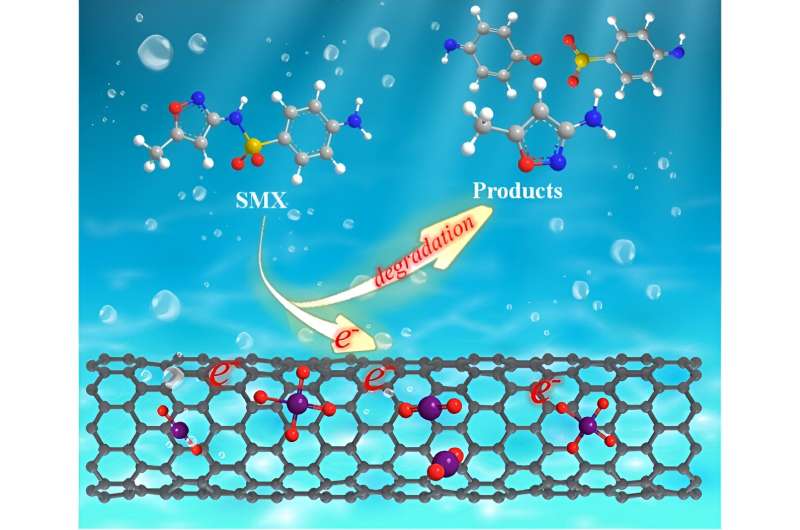
CNT is an excellent electron transfer mediator, had been proved as a 'bridge' to facilitate the electron delivery from organic molecules (electron donor) to persulfate (electron acceptors). This may lead to oxidative decomposition of organic contaminants (OCs), rather than converting from KMnO4 to reactive manganese species.
In order to overcome the mass transfer limitation, researchers from Donghua University and Harbin Institute of Technology designed and established a flow-through KMnO4/CNT system.
This study titled "Insights into the electron transfer mechanisms of permanganate activation by carbon nanotube membrane for enhanced micropollutants degradation" was published online in Frontiers of Environmental Science & Engineering .
In this study, the research team designed a catalytic CNT membrane for KMnO4 activation toward enhanced degradation of micropollutants. The treatment effect of the system was optimized by selecting appropriate operating parameters.
Analysis of experimental data and theoretical calculations revealed the reaction mechanism and compared the utilization efficiency of permanganate in different systems. In addition, using advanced analytical methods, the degradation pathways of the target substances were revealed and the toxicity of the intermediates was evaluated.
Their results revealed that the flow-through KMnO4/CNT system outperformed conventional batch reactor. Under optimal conditionals, a > 70% removal (equivalent to an oxidation flux of 2.43 mmol/[h·m2]) of 80 μmol/L sulfamethoxazole (SMX) solution can be achieved at single-pass mode.
The experimental analysis and DFT studies verified that CNT could mediate direct electron transfer from organic molecules to KMnO4, resulting in a high utilization efficiency of KMnO4.
Furthermore, the KMnO4/CNT system had outstanding reusability and CNT could maintain a long-lasting reactivity, which served as a green strategy for the remediation of micropollutants in a sustainable manner. This study not only demonstrated the potential application of CNT as electronic media in advanced oxidation processes. Moreover, the system design was robust and efficient, and provided a new solution for green environment remediation.
More information: Xufang Wang et al, Insights into the electron transfer mechanisms of permanganate activation by carbon nanotube membrane for enhanced micropollutants degradation, Frontiers of Environmental Science & Engineering (2023). DOI: 10.1007/s11783-023-1706-0
Provided by Higher Education Press