This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
proofread
Recent advances in nickel-based catalysts for electrochemical reduction of carbon dioxide
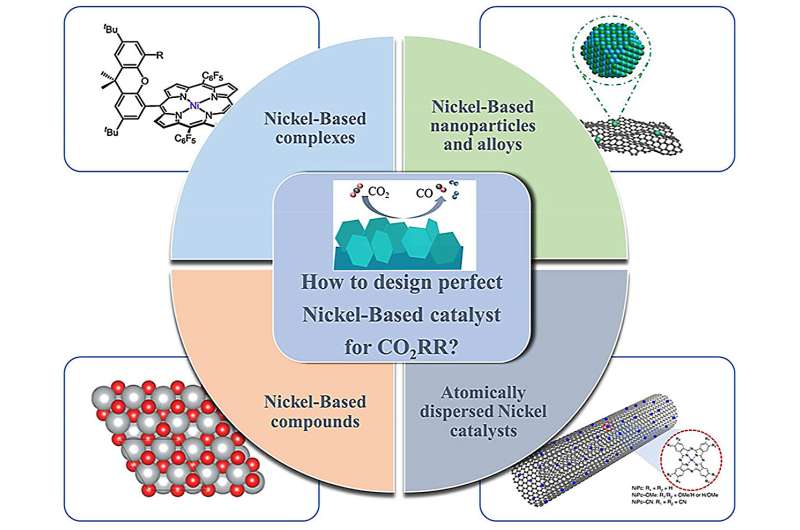
In a paper published in Advanced Sensor and Energy Materials, an international team of scientists reviewed recent advances in nickel-based catalysts for electrochemical reduction of carbon dioxide. The relevant CO2RR mechanisms are first discussed in detail.
Following this, several typical Ni-based catalysts including Ni-based complexes catalysts, nanoparticles and alloys, Ni-based compounds, and atomically dispersed Ni catalysts are summarized. The preparation strategies for industrial-level electrocatalysts are also highlighted in this review.
Finally, the opportunities, challenges, and future outlooks of CO2RR technology are summarized. This review introduces the recent advances and frontiers of nickel-based CO2RR catalysts, which enlightens the path for further exploration of highly efficient CO2RR electrocatalysts. This study is led by Meiling Xiao from State Key Laboratory of Electroanalytical Chemistry, Changchun Institute of Applied Chemistry.
The study is led by Meiling Xiao (State Key Laboratory of Electroanalytical Chemistry, Changchun Institute of Applied Chemistry, China).
Carbon dioxide is a notorious greenhouse gas, contributing to energy scarcity and the greenhouse effect. By employing techniques for direct capture, conversion, and utilization, it is possible to effectively reduce the levels of carbon dioxide in the atmosphere. CO2 can be transformed into valuable materials through various methods, including electrochemistry, photochemistry, biochemistry, and thermochemistry.
Because of the ability to store intermittent renewable energy with nearly zero emissions, electrochemical CO2 reduction is an intriguing field. Meanwhile, CO2 reduction reaction (CO2RR) stands out due to its high reaction rate and its flexible and controllable process, making it the most promising avenue for industrial-scale renewable energy storage.
Despite considerable efforts, the activation of inert linear CO2 molecules remains a significant challenge due to thermodynamic stability and sluggish reaction kinetics, thereby impacting the CO2 reduction reaction.
On the other hand, a wide range of possible products can be formed during practical CO2 reduction processes, such as carbon monoxide, formate, methanol, ethanol, ethylene, and hydrocarbons, reflecting the diverse reaction pathways of CO2 reduction that exhibit significant variations under different conditions.
Given the fact that many reaction pathways have similar theoretical initiation potentials, simultaneous production of different products can occur during the reaction, resulting in poor selectivity. Moreover, the competitive hydrogen evolution reaction (HER) also leads to low Faradaic efficiencies (FE) for specific products.
Therefore, the exploration of catalysts suitable for CO2 reduction is of utmost importance. Since the pioneering work by Hori and colleagues in 1985 on electroreduction products, researchers have tested various metallic materials. Over the past few decades, precious metals like gold, platinum, silver, and palladium have exhibited excellent CO2RR performance.
However, their low abundance and high cost pose challenges for industrial implementation. Hence, non-precious metals with satisfactory selectivity and high FE have been extensively studied as low-cost catalysts. These electrode materials include a wide range of metals such as copper, zinc, tin, bismuth, and nickel.
Among these catalysts with metal atoms as active centers, catalytic materials with Ni sites exhibit more prominent advantages. As a popular earth-abundant metal belonging to Group VIII-B, Ni is regarded as an ideal substituent for precious metals in the design of CO2RR catalysts, due to a wide variety of Ni valences from 0 to +4.
The rich redox properties coupled with various coordination geometries make rational design of ligand frameworks possible to coordinate with the Ni center, imparting specific catalytic functions to nickel-based materials. Additionally, Nickel has been identified as an active center in natural enzymes, exemplified by the renowned CO dehydrogenases (CODHs), which facilitate the reversible conversion between CO2 and CO.
Both metallic and oxidized Ni can selectively convert CO2 to C1 products at remarkably high rates, while suppressing HER. Ni single-atom catalysts (SACs) exhibit higher reactivity compared to other earth-abundant metals, owing to their moderate CO2 adsorption capability and d-band proximity to the Fermi level. Ni SACs have been explored as efficient cathode materials for CO2RR, demonstrating high activity and selectivity.
Moreover, Ni is one of the few metals capable of efficiently catalyzing the reduction of CO2 into hydrocarbons and alcohols. The uniqueness of Ni-based catalysts in the CO2RR process can be attributed to the moderate combination between ∗CO and the Ni-based catalyst, facilitating the activation of CO2 and the subsequent hydrogenation of ∗CO, which is a crucial step in hydrocarbon formation.
For example, Ni–Ga alloys were capable of producing ethylene and ethane. Ni phosphides can selectively reduce CO2 to C3 and C4 products (methylglyoxal and 2,3-furandiol, respectively) with a low overpotential (10 mV) and current densities. The unexpected ability of Ni-based electrocatalysts to promote C–C coupling reactions is noteworthy, considering bulk Ni susceptibility to CO poisoning.
Strong interactions with CO can lead to deactivation or blocking of active sites. Hence, understanding the deactivation mechanisms is crucial. In addition to catalyst design, recent advancements have also focused on improving catalytic systems. Gas diffusion electrodes (GDEs) have been widely employed to directly transport reactant gases to the interface between the catalyst and the electrolyte, enhancing the CO2 concentration at the catalyst surface and promoting current density.
This review summarizes the recent advancements in efficient Ni-based catalysts and CO2RR systems. Firstly, the review provides a brief introduction to the reaction mechanisms responsible for the production of C1 and C2+ products.
The main focus is on recent developments specifically related to Ni-based catalysts, including Ni-based complexes, Ni-based single/multi-metallic catalysts, Ni-based compounds, and atomically dispersed catalysts.
Factors that influence catalytic efficiency, such as catalyst surface structure, catalyst size, and surface state, are described in detail. Subsequently, the review outlines strategies concerning catalyst and system design for achieving high Faradaic efficiency (FE) and current density. Finally, the review presents an overview of future opportunities and challenges in the field.
More information: Xiao-Hui Liu et al, Recent advances in nickel-based catalysts for electrochemical reduction of carbon dioxide, Advanced Sensor and Energy Materials (2023). DOI: 10.1016/j.asems.2023.100073
Provided by Beijing Zhongke Journal Publising Co.