This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Structures of connexin-43 gap junction channel and hemichannel in a putative closed state
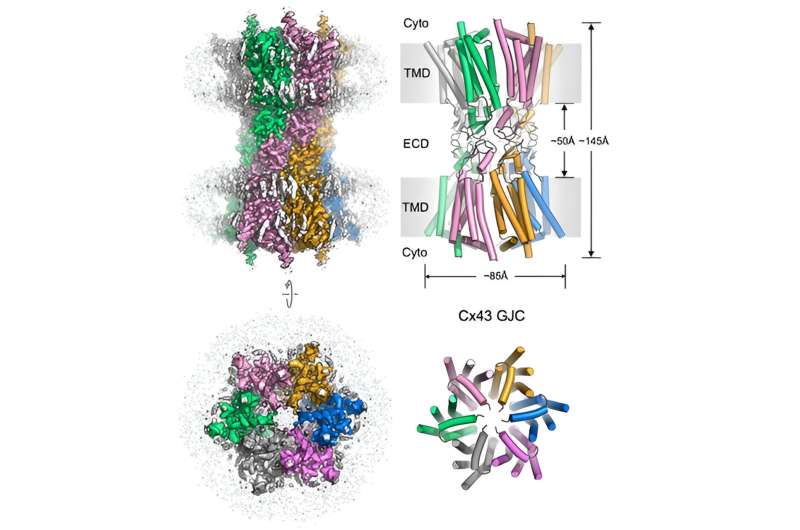
A recent eLife paper by the group of Volodymyr Korkhov (IMBB, ETHZ & PSI), in collaboration with the Francesco Gervasio (UniGe), Mario Bortolozzi (UniPD), Paola Picotti (IMSB, ETHZ) and Nicola Zamboni (IMSB, ETHZ) groups describes the structures of connexin-43 gap junction channel and hemichannel in a putative closed state.
Gap junction channels (GJCs) mediate intercellular communication by connecting two neighboring cells and enabling direct exchange of ions and small molecules. Cell coupling via connexin-43 (Cx43) GJCs is important in a wide range of cellular processes in health and disease, yet the structural basis of Cx43 function and regulation has not been determined until recently.
The team described the structure of a human Cx43 GJC determined by cryo-EM and single particle analysis at 2.26 Å resolution. The pore region of Cx43 GJC features several lipid-like densities per Cx43 monomer, located close to a putative lateral access site at the monomer boundary.
The researchers found a previously undescribed conformation on the cytosolic side of the pore, formed by the N-terminal domain and the transmembrane helix 2 of Cx43 and stabilized by a small molecule. Structures of the Cx43 GJC and hemichannels (HCs) in nanodiscs revealed a similar gate arrangement. The features of the Cx43 GJC and HC cryo-EM maps and the channel properties revealed by molecular dynamics simulations suggested that the captured states of Cx43 may correspond to a novel conformation of the channel, a closed state.
More information: Chao Qi et al, Structure of the connexin-43 gap junction channel in a putative closed state, eLife (2023). DOI: 10.7554/eLife.87616
Journal information: eLife
Provided by ETH Zurich