This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Scientists demonstrate 3D 'bio-printing' inside organoids growing in hydrogels
Scientists from the NIHR Great Ormond Street Hospital Biomedical Research Centre (a collaboration between GOSH and UCL), London, and University of Padova, Italy, have shown for the first time how 3D printing can be achieved inside "mini-organs" growing in hydrogels—controlling their shape, activity, and even forcing tissue to grow into "molds."
This can help teams study cells and organs more accurately, create realistic models of organs and disease, and even better understand how cancer spreads through different tissues.
A particularly promising area of research at the Zayed Centre for Research (a partnership between Great Ormond Street Hospital (GOSH), GOSH Charity and University College London Great Ormond Street Institute of Child Health (UCL GOS ICH)) is organoid science—the creation of micro-versions of organs like the stomach, the intestines and the lungs.
But this tissue almost always grows in an uncontrolled way and doesn't represent the complex structure of naturally occurring organs. This is particularly important as an organ's shape and structure is as crucial as its cellular make up—in the stomach, or lungs and heart, for example.
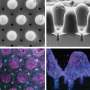
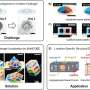
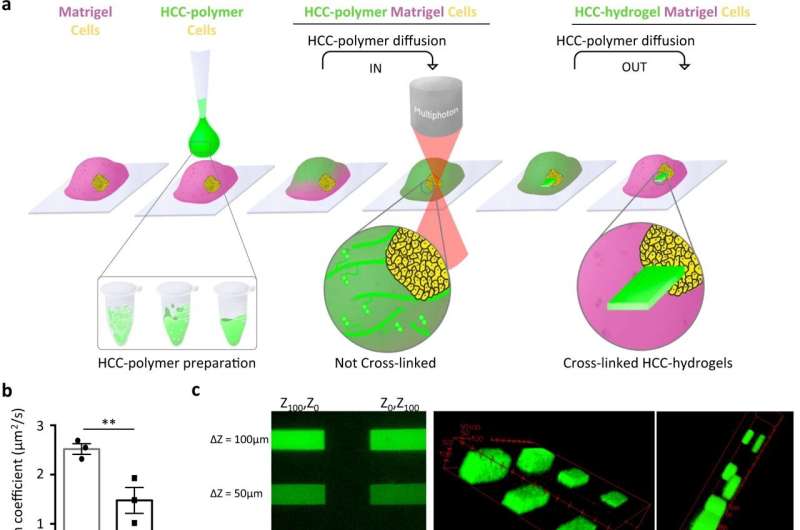
This research shows, how scientists can create solid structures within a pre-existing gel to solidify specific patterns in real time, guiding organoids growing in the gel into a particular structure by using light from a high-specification microscope. This means that any cell in the growing mini-organ or entire organoids will grow in a specific and precise way.
The paper, published in Nature Communications, shows how the team hope to recreate and study what happens to an organ's function when it doesn't grow correctly—for example in many malformations that develop in the early stages of pregnancy.
The team hope this research will create better models of disease which means that their studies are more reliable, the results are of better quality and the need for animal research is one day reduced. The work could also lead to treatment through the delivery of biologically accurate 'patches' in living organs.
Examples of 'printing' uses:
- Ordered cells: To study microscopic brain cells called neurons, organoid research would traditionally create disordered bundles of neurons that are impossible to isolate and study. This technique allows the team to create hardened gel 'rails' for the neurons to grow along, like the lanes of an Olympic pool.
- Defined shapes: To create microscopic intestines with the same shape as 'real' developing intestines, the team created a complex hydrogel mold that guided organoids into shapes that mimic the complex structures of a developing intestine called 'crypts' and 'villi."
- Creating branches: Scientists were able to pattern a hydrogel to encourage lung cells to create branches, like they do in 'real' lung.
- Cancer spread: To study how cancer travels through tissue of different hardness and density, the team created hardened gel cages around cancer cells and monitored how the cancer cells movement changes depending on the density of its surroundings—this is important for understanding the spread of cancer
Dr. Giovanni Giobbe from UCL GOS ICH, Co-lead author of the research said, "It's been amazing to see these precise structures begin to form before our eyes due to our small but painstaking adjustments in the polymer gel. We're really excited to see where this can take us in the understanding of human disease and one day, treatment."
Dr. Anna Urciuolo from University of Padova and lead of the Neuromuscular Engineering Lab at the Institute of Pediatric Research said, "This work is an exemplar of the advancements of the multidisciplinary approach that is exploding in biomedical research. The ability to reproduce models of organs in the lab and the development of technologies that help scientist to recapitulate healthy and diseased tissues and organ complexity on the bench is the outset of how translational medicine will change in the next future/"
Professor Paolo De Coppi, Pediatric surgeon at GOSH, Professor of Pediatric Surgery at UCL GOS ICH, and co-lead of the tissue engineering and regenerative medicine theme at the NIHR GOSH BRC said, "This work as an excellent example of how we can bring interdisciplinary, international teams together to improve our research and benefit patients."
"Teams at GOSH and UCL that specialize in organoid research in the UK, working with Italian teams specializing in design and application of gel-printing, are what have made this incredible and beautiful piece of research come to fruition. This will have implications for laboratory-based research to improve our understanding of disease but could also lead to in-patient uses and treatments."
Next steps for this work will be to study these controlled, molded and directed mini-organs to better understand how they can mimic real organs and conditions in the body.
More information: Anna Urciuolo et al, Hydrogel-in-hydrogel live bioprinting for guidance and control of organoids and organotypic cultures, Nature Communications (2023). DOI: 10.1038/s41467-023-37953-4
Journal information: Nature Communications
Provided by University College London