This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
trusted source
proofread
Emergence of solvated dielectrons observed for the first time
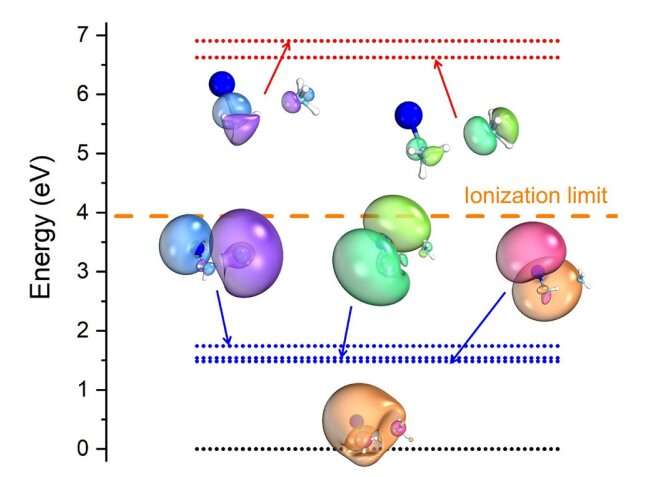
Solvated dielectrons are the subject of many hypotheses among scientists, but have never been directly observed. They are described as a pair of electrons that is dissolved in liquids such as water or liquid ammonia. To make space for the electrons a cavity forms in the liquid that the two electrons occupy.
An international research team led by Dr. Sebastian Hartweg, initially at Synchrotron SOLEIL (France), now at the Institute of Physics at the University of Freiburg and Prof. Dr. Ruth Signorell from ETH Zurich, including scientists from the synchrotron SOLEIL and Auburn University has now succeeded in discovering a formation and decay process of the solvated dielectron.
In experiments at the synchrotron SOLEIL (DESIRS beamline), the consortium found direct evidence supported by quantum chemical calculations for the formation of these electron pairs by excitation with ultraviolet light in tiny ammonia droplets containing a single sodium atom. The results were recently published in the journal Science.
Traces of an unusual process
When dielectrons are formed by excitation with ultraviolet light in tiny ammonia droplets containing a sodium atom, they leave traces in an unusual process that scientists have now been able to observe for the first time. In this process, one of the two electrons migrates to the neighboring solvent molecules, while at the same time the other electron is ejected. "The surprising thing about this is that similar processes have previously been observed mainly at much higher excitation energies," says Hartweg.
The team focused on this second electron because there could be interesting applications for it. On the one hand, the ejected electron is produced with very low kinetic energy, so it moves very slowly. On the other hand, this energy can be controlled by the irradiated UV light, which starts the whole process. Solvated dielectrons could thus serve as a good source of low-energy electrons.
Generated specifically with variable energy
Such slow electrons can set a wide variety of chemical processes in motion. For example, they play a role in the cascade of processes that lead to radiation damage in biological tissue. They are also important in synthetic chemistry, where they serve as effective reducing agents.
By being able to selectively generate slow electrons with variable energy, the mechanisms of such chemical processes can be studied in more detail in the future. In addition, the energy made available to the electrons in a controlled manner might also be used to increase the effectiveness of reduction reactions.
"These are interesting prospects for possible applications in the future," says Hartweg. "Our work provides the basis for this and helps to understand these exotic and still enigmatic solvated dielectrons a little better."
More information: Sebastian Hartweg et al, Solvated dielectrons from optical excitation: An effective source of low-energy electrons, Science (2023). DOI: 10.1126/science.adh0184
Journal information: Science
Provided by Albert Ludwigs University of Freiburg