This article has been reviewed according to Science X's editorial process and policies. Editors have highlighted the following attributes while ensuring the content's credibility:
fact-checked
peer-reviewed publication
proofread
Advancing our mastery of stereoselective photoredox reactions to produce mirror-image molecules

Researchers of the Max-Planck-Institut für Kohlenforschung have expanded the molecular toolbox for efficient, targeted syntheses: To do so, they use a very special catalyst—and the energy of light. The result of their work has now been published in Science.
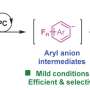
Typically, in a so-called photoredox reaction, light is absorbed by a photocatalyst. A single electron transfer then occurs between the catalyst and a substrate, creating a radical ion, which is highly reactive and can engage in various desirable transformations.
The problem with these "radicals," however, has been to control their selectivity. A second catalytic activation mode was often necessary to obtain a desired product in the required purity. This has previously limited stereoselective photoredox reactions to special substrates amenable to that second activation mode.
"Photocatalysis enables chemical reactions to take place with the help of light—for example, in the leaves of plants, but also in the production of drugs," explains Benjamin List, director at the Max-Planck-Institute. Photocatalytic reactions proceed via high-energy intermediates, and controlling the selectivity of their reactions has been difficult. "We now provide a general concept for carrying out these reactions with high stereoselectivity, in which we can produce mirror-image molecules," says Benjamin List.
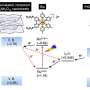
This means that the photocatalyst absorbs light and takes up an electron from the substrate, i.e. a reaction partner. Since this electron is negatively charged, the substrate now automatically becomes positive and combines with a counteranion. This ion pair is now involved in another reaction step—the one that really interests the scientists. The original photocatalyst instead is no longer part of this step—and can be regenerated and used again to absorb light and pick up an electron.
With this new method, the researchers of the Kohlenforschung have succeeded in developing another molecular precision tool. "We haven't really thought about specific applications yet," says Dr. Sayantani Das, postdoctoral researcher with Benjamin List and involved in the project. But asymmetric photoredox catalysis could certainly be useful in the field of synthesis—for example, in the production of drugs or fragrances.
More information: Sayantani Das et al, Asymmetric counteranion-directed photoredox catalysis, Science (2023). DOI: 10.1126/science.ade8190
Journal information: Science
Provided by Max-Planck-Institut für Kohlenforschung