A novel gene in mammals that controls a new structure found in nerve cells
Evolution is often portrayed as a "tinkering" process, one that makes use of slight modifications to pre-existing capabilities. So how do organisms evolve brand new structures?
A new study by Dr. Zsuzsanna Izsvák from the Max Delbrück Center for Molecular Medicine in the Helmholtz Association (Max Delbrück Center) and Professor Laurence Hurst from the Milner Centre for Evolution at University of Bath (UK) found evidence that evolution of a new gene underpins the evolution of a new structure found in nerve cells. They describe this unusual gene called piggyBac Transposable Element-derived 1, or PGBD1, in the journal Molecular Biology and Evolution.
'Jumping genes' cause mutations
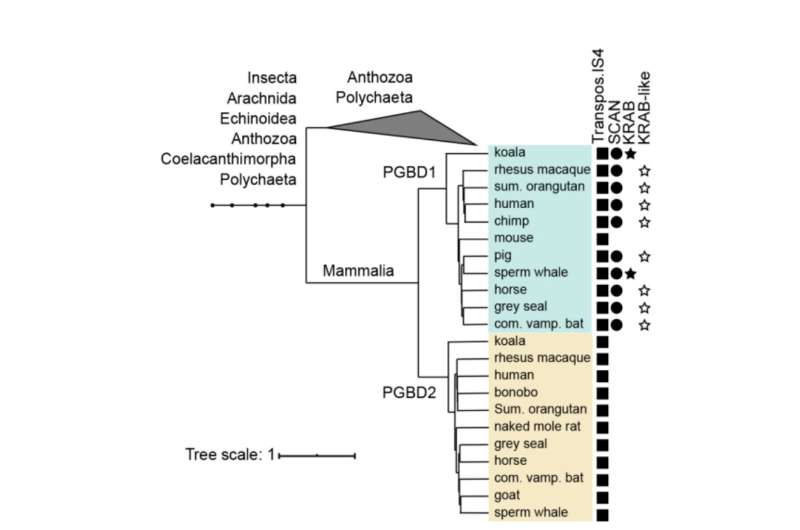
PGBD1 is one of five related PGBD genes that shows a distinct resemblance to the piggyBac element first identified in insects—hence the name piggyBac Transposable Element-derived. The PiggyBac elements are "jumping genes", also called transposons. They are able to copy themselves and to move from one location in the genome to another, sometimes introducing mutations or changing functions. PiggyBac transposons arrived into our species by horizontal transfer—similar to how some viruses can integrate their genome into our DNA.
However, while the piggyBac transposons have lost their ability to jump around in our DNA over time, five piggyBac Transposable Element-derived genes (PGBD1-5) have been fixed in humans. "We aimed at finding out what potentially useful function the PGBD genes might have," says Zsuzsanna Izsvák. "For this study, we focused on PGBD1."
Among the five PGBD genes, PGBD1 is unique in that it has also incorporated parts of other genes, resulting in a protein that has extra parts that are able to bind other proteins and to bind DNA. PGBD1 is thus a novel gene that is part human gene fragment, part inactive jumping gene.
PGBD1 regulates nerve cells and their 'protein traps'
PGBD1 is found only in mammals. It is particularly active in cells that become neurons. The researchers first investigated, where PGBD1 protein binds to DNA, observing that it glues itself in and around genes associated with nerve development. They found PGBD1 controls nerve cell development by blocking genes expressed in mature nerve cells while keeping those genes associated with being pre-nerve cells activated. Reducing the level of PGBD1 in pre-nerve cells caused them to start developing as nerve cells.
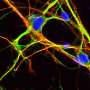
One of the genes that PGBD1 protein binds especially attracted their interest. NEAT1 is a strange gene that codes for an RNA which, unusually, doesn't then go on to make a protein. Instead, this product, a non-coding RNA, makes the backbone of a physical structure, the paraspeckles. These are tiny structures in the nuclei of some of our cells that act like traps for some RNAs and proteins.
The researchers found that in pre-nerve cells PGBD1 protein binds to the NEAT1 gene and stops it from working. However, when PGBD1 levels go down, NEAT1 RNA levels go up, paraspeckles form and cells become mature nerve cells. PGBD1 thus has evolved to be a key regulator of presence or absence of paraspeckles, and thus the regulator of nerve cell development.
Evolution is not random tinkering
What, however, is most intriguing is that paraspeckles are, like PGBD1, also mammal-specific. PGBD1 is then a rare example of a new gene that has evolved to regulate a new structure, albeit a rather small one. Zsuzsanna Izsvák, co-senior author from Max Delbrück Center, says, "This is a really unusual and serendipitous discovery. We have known that duplication of pre-existing genes can underpin the evolution of novelty, but this is a rare example of evolution doing more than just tinkering. This is a novel gene to control a novel structure." The exciting question now is whether it also plays a role in adult neurons.
Co-senior author Professor Laurence Hurst of the Milner Centre for Evolution at the University of Bath adds that they "have worked out how paraspeckles are controlled, now we just need to work out how the paraspeckle itself evolved. This might be a much harder task as non-coding RNAs like NEAT1 tend to be fast evolving and therefore hard to trace over evolutionary time."
This coupling between NEAT1 and PGBD1 may also be involved in schizophrenia. While NEAT1 has been previously associated with this neurological disease, the team identified some mutations in PGBD1 that they could show were also common in patients with schizophrenia—one of these mutations changes the protein of PGBD1 while others may control its level.
First author Dr. Tamas Raskó, at the time of the study a postdoctoral researcher in the group of Zsuzsanna Izsvák says that "it is surely more than coincidence that both genes are involved in schizophrenia. It is very unusual to find a mutation that changes a protein that is coupled to this disease. The effects of this mutation must be a priority for further studies."
More information: Tamas Rasko et al, A novel gene controls a new structure: PiggyBac Transposable Element-derived 1, unique to mammals, controls mammal-specific neuronal paraspeckles, Molecular Biology and Evolution (2022). DOI: 10.1093/molbev/msac175
Journal information: Molecular Biology and Evolution
Provided by Max Delbrück Center for Molecular Medicine