Selective cancer nanoparticle targeting under the microscope
Nanoparticles can be used as powerful vehicles to administer vaccines and prevent serious illness, as with the treatment of COVID-19 and to deliver chemotherapeutic drugs to cancer cells with goal of eradicating the cancer cells and leaving the healthy cells unharmed. For cancer patients, this has the potential to reduce severe side effects that originate from the toxicity of chemotherapeutics. Unfortunately, no clinically applied selective nanoparticle treatment (also known as nanotherapeutic) exists yet, with research focusing on improving and understanding current nanotherapeutics. For her Ph.D. research, Laura Woythe took a closer look at nanoparticles and cancer cells to design selective nanotherapeutics using advanced optical microscopy techniques.
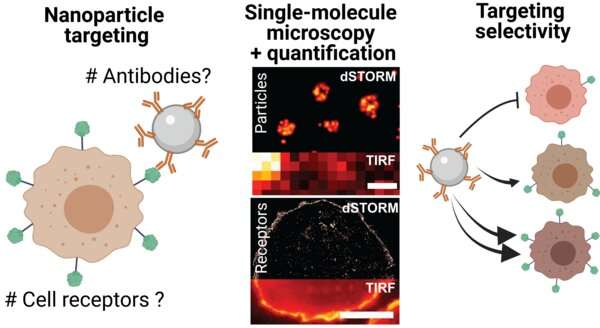
To enhance the ability of nanoparticles to target cancer cells, scientists can exploit how nanoparticles interact with specific cell biomarkers or "receptors" on the surface of cells. For this purpose, molecules or "ligands" that recognize specific cell receptors are placed on the surface of the nanoparticles.
However, this so-called functionalization process is hard to control, due to the small size of the nanoparticles, causing some molecules to be misplaced, to function incorrectly, or to be erroneously attached on the nanoparticle surface. All of these reduce a nanoparticle's capacity to interact with cancer cells in the intended way.
Furthermore, questions remain regarding the efficiency of such attachment protocols, and if the number of molecules we are attaching is efficient enough to target cancer cells. The challenges lie in the small size of the molecules and cell receptors, and the limited quantitative methods that are available to estimate the number of molecules on the surface of the nanoparticles. In other words, how can scientists count the number of molecules on the surface of the nanoparticles in order to check that the nanoparticles can be effective against cancer cells?
Super-resolution microscopy
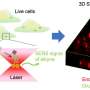
For her Ph.D. research, Laura Woythe investigated the functionality of molecules attached to nanoparticles for selective cancer targeting using advanced optical microscopy techniques or "super-resolution" optical microscopy.
Super-resolution microscopy encompasses a group of microscopy techniques that have a resolution power that is 10 times higher than conventional optical microscopy. This allows for the visualization of nanometric structures, such as nanoparticles and cell receptors, in the range of 10 to 100 nanometers (nm). This size range is equivalent to visualizing structures up to 5,000 times smaller than a human hair.
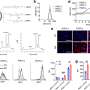
Using super-resolution microscopy, Woythe and her colleagues were able to count individual ligands on nanoparticles and receptors on cancer cells, thus enabling the finetuning of the targeting interaction. These numbers can go a long way towards developing more efficient nanotherapeutic delivery.
Woythe's research is an important step towards a better understanding of nanomaterials for biomedical applications, specifically selective cell targeting of cancer cells and diseased cells without affecting healthy tissue, and thus minimizing the potential side-effects and the burden this cause to cancer patients.
More information: Molecular Mapping of Nanoparticle Targeting: A Super-Resolved Journey. research.tue.nl/en/publication … -super-resolved-jour
Laura Woythe et al, A Single-Molecule View at Nanoparticle Targeting Selectivity: Correlating Ligand Functionality and Cell Receptor Density, ACS Nano (2022). DOI: 10.1021/acsnano.1c08277
Journal information: ACS Nano
Provided by Eindhoven University of Technology