Interaction between nanoplastics and pectin, a water-soluble polysaccharide
Microplastics are known to collect in ecosystems and nanoplastics occur from the breaking down of microplastics. Nanoplastics are plastic particles of sizes less than 100 nm and when they are in water, they are dispersed in a colloidal form. Nanoplastics might be more prevalent than microplastics, but it is hard to analyze and study them in-depth due to their size. In zebrafish, however, nanoplastics have been found in various organs including the brain, which may be an indicator that it crosses the blood-brain barrier.
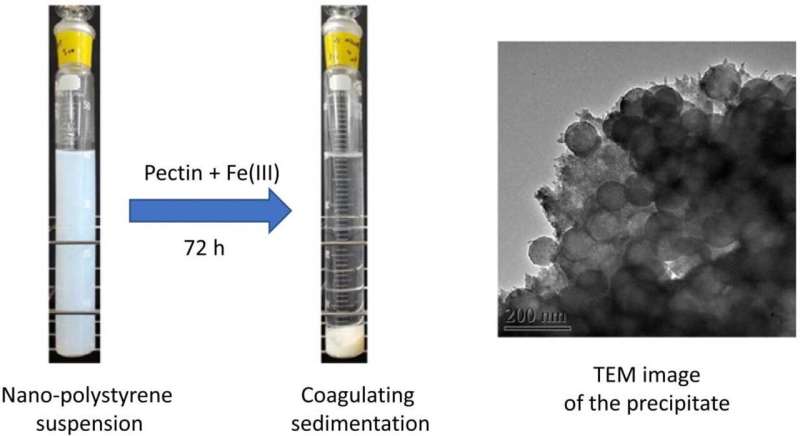
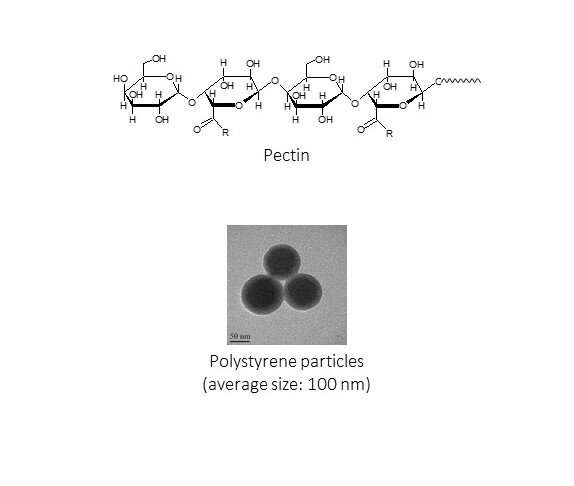
In towns and cities, 90% of microplastics are captured in the sewage treatment process. In the ocean, microplastics are also known to sink to the bottom by binding to biopolymers. Therefore, a research team at Shinshu University led by Professor Hiroshi Moriwaki of the Department of Applied Biology, Faculty of Textile Science and Technology considered using pectin, a biopolymer to bind to nanoplastics with the help of Fe (III) or AI (III). They found that they were able to remove 95% of nanoplastics in the first 24 hours by using coagulating sedimentation with pectin and Fe(III) with filter paper.
The use of pectin was inspired by the abundance of apples in the prefecture of Nagano where Shinshu University is based. The research was published in the Journal of Environmental Chemical Engineering.
More information: Hiroshi Moriwaki et al, Interaction between nanoplastics and pectin, a water-soluble polysaccharide, in the presence of Fe(III) ion, Journal of Environmental Chemical Engineering (2022). DOI: 10.1016/j.jece.2022.108054
Provided by Shinshu University