Transcriptional regulation mechanism of hard clams against heat and hypoxia stress
Temperature and dissolved oxygen (DO) can affect a series of physiological and biochemical responses and drive the expression of regulatory genes. When water temperature increases, the solubility of oxygen decreases, leading to hypoxia.
The hard clam Mercenaria mercenaria is a heat- and hypoxia-tolerant species, which is an ideal model for investigating the mechanisms underlying environmental adaptation.
Recently, a research team led by Prof. Zhang Tao from the Institute of Oceanology of the Chinese Academy of Sciences (IOCAS) has explored the transcriptional regulation mechanism of hard clam under heat, hypoxia and combined stress.
The study was published in Aquaculture.
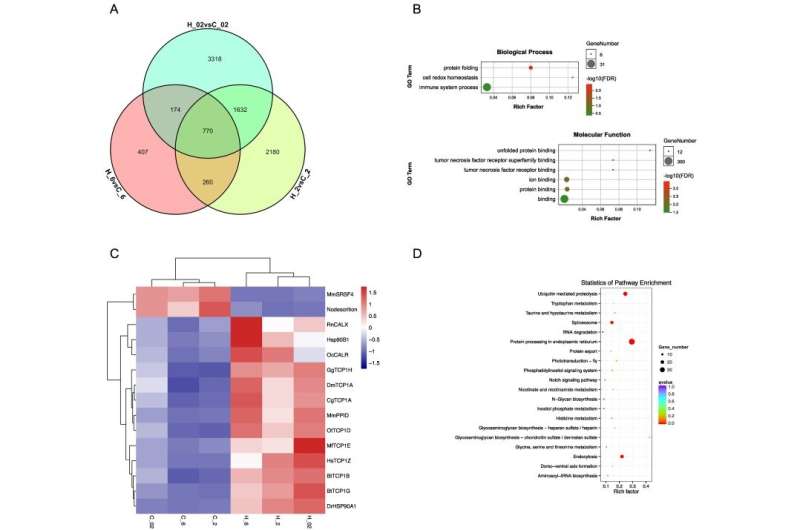
Protein folding plays a vital role in protein homeostasis. The researchers found that it was significantly enriched in common differentially expressed heat-induced genes. Heat stress could destroy protein structures, and molecular chaperones including HSP90 and TCP1 exhibited high expression in the heat-stress groups. Molecular chaperones assist in protein folding to ensure that protein homeostasis maintained in cells.
Microtubule-related GO terms were significantly enriched in hypoxia-induced genes under different temperatures. Microtubules are very important in forming the cytoskeleton and maintaining the cell structure and motility.
Kinesin and dynein-related genes were upregulated in the hypoxia groups compared to the control. Dynein-mediated microtubule motor and movement might help cytoskeletal reorganization and maintain cell structure in hard clams under hypoxia stress.
Protein processing in endoplasmic reticulum and ubiquitin-mediated proteolysis were activated under heat plus hypoxia stress. Hard clams might employ a strict quality control system to alleviate cytotoxicity and maintain cell homeostasis by ensuring proper protein folding, enhancing recognition of misfolded protein, and facilitating damaged protein degradation through ubiquitin-mediated proteolysis.
More information: Zhi Hu et al, Mechanisms of heat and hypoxia defense in hard clam: Insights from transcriptome analysis, Aquaculture (2021). DOI: 10.1016/j.aquaculture.2021.737792
Provided by Chinese Academy of Sciences