Light-independent metabolic pathways regulate astaxanthin accumulation in Haematococcus
A research team led by Prof. Liu Jianguo from the Institute of Oceanology of the Chinese Academy of Sciences found that light-independent metabolic pathways, in addition to light-dependent metabolic pathways, can also regulate astaxanthin accumulation in Haematococcus.
Their findings were published in Bioresource Technology.
Astaxanthin has multiple application prospects in nutraceuticals, pharmaceuticals and cosmetics owing to its strong colorant, antioxidant capacity and other biological functions. H. pluvialis, a unicellular green alga rich in astaxanthin, is recognized as the best biological resource for producing natural astaxanthin.
Traditionally, astaxanthin biosynthesis in H. pluvialis was considered to be inducted mainly by both high light and/or nutrient deficiency. Therefore, most previous studies on the regulation of astaxanthin biosynthesis and accumulation have been focused on the light-dependent metabolic pathways, such as photosynthesis and photoprotection. There are few reports on the role of light-independent respiratory pathway during astaxanthin accumulation.
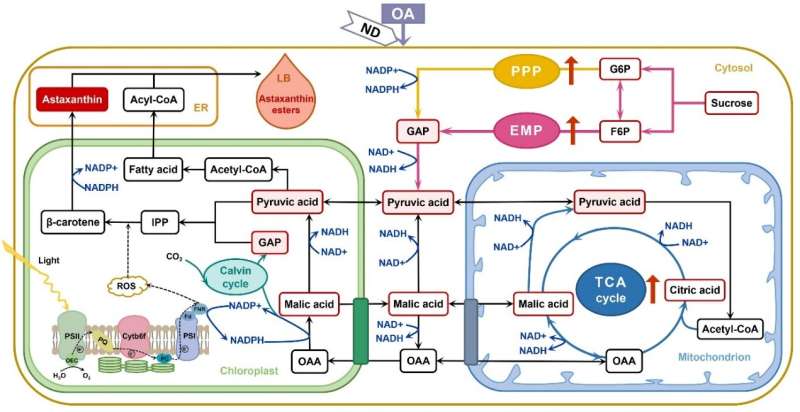
In their studies, the researchers found that light-independent metabolic pathways, such as the Embden-Meyerhof-Parnas pathway, tricarboxylic acid cycle, pentose phosphate pathway and alternative oxidase respiratory pathway, also play important roles in regulating astaxanthin accumulation in Haematococcus.
"The activity of mitochondrial alternate oxidase pathway has significant negative correlation with astaxanthin accumulation in H. pluvialis," said Prof. Liu. Based on this discovery, a new viewpoint that regulating respiration would promote astaxanthin biosynthesis in H. pluvialis was firstly proposed in the study published on Nov. 6.
The researchers also investigated the effects of exogenous respiratory metabolic intermediates on astaxanthin synthesis, and further proved that tricarboxylic acid cycle can provide carbon skeletons for astaxanthin biosynthesis in H. pluvialis.
In their another study published in August, they found exogenous fumarate, a TCA cycle intermediate, enhanced the activities of EMP, PPP and TCA respiratory metabolic pathways, resulting in the significantly increased level of pyruvate and glyceraldehyde-3-phosphate. On the one hand, the enhanced substrate pools could promote the generation of astaxanthin precursor, isopentenyl pyrophosphate (IPP), and then directly facilitate astaxanthin synthesis. On the other hand, the accumulation of fatty acids could facilitate astaxanthin synthesis indirectly by promoting the process of astaxanthin esterification.
The researchers also found exogenous oxaloacetate, another TCA cycle intermediate, would promote astaxanthin biosynthesis by improving the levels of substrates and NADPH. Moreover, oxaloacetate had a faster and more obvious effect than fumarate, which was mainly due to its more efficient transmembrane mechanism, according to the study published on Dec. 4.
The work will help to complete the H. pluvialis development model based on the cell cycle control and large-scale closed photobioreactor construction.
More information: Wenjie Yu et al, Enhancement of astaxanthin accumulation in Haematococcus pluvialis by exogenous oxaloacetate combined with nitrogen deficiency, Bioresource Technology (2021). DOI: 10.1016/j.biortech.2021.126484
Litao Zhang et al, A strategy for promoting carbon flux into fatty acid and astaxanthin biosynthesis by inhibiting the alternative oxidase respiratory pathway in Haematococcus pluvialis, Bioresource Technology (2021). DOI: 10.1016/j.biortech.2021.126275
Wenjie Yu et al, Exogenous sodium fumarate enhances astaxanthin accumulation in Haematococcus pluvialis by enhancing the respiratory metabolic pathway, Bioresource Technology (2021). DOI: 10.1016/j.biortech.2021.125788
Journal information: Bioresource Technology
Provided by Chinese Academy of Sciences