Assembling materials using charged polymers
Soft matter can be found in many everyday materials, such as food, cosmetics, and the cells in our own bodies. The structure of these tiny, soft materials is determined by various interactions, one of which is electrostatic interaction. For his Ph.D. research, Christian Sproncken used electrostatic interactions to assemble soft materials into various structures consisting of charged polymer chains. These materials could be used in responsive coatings for photonic microchips, or in materials to stop ice growth on car windshields or airplane wings.
Molecules of opposite charge attract each other, and this attraction can be used to build materials with interesting properties. Underpinning this attraction is electrostatic interaction, which is not only reversible but also tunable by varying solution properties such as salt concentration or pH.
Using two polymers with opposite charges, various materials can be made by controlling how the polymers co-assemble via electrostatic interaction. For Christian Sproncken, the primary goal of his Ph.D. research was to design and study the properties of materials made from oppositely charged polymers.
The world is soft
"The world is full of soft materials. Just think of long-chain polymers in cosmetics or cells in the body. Of course, these structures are built from smaller units, like emulsions or short-chain polymers," says Sproncken, who completed his research in the Laboratory of Self-Organizing Soft Matter led by Ilja Voets. "But what's really interesting is that these units can be put together in different ways to make new materials."
In his research, Sproncken was mainly interested in combining oppositely charged polymers to assemble new materials that could be used for various applications such as coatings in reprogrammable photonic materials and for materials with anti-freeze abilities.
The various studies, versatile approaches, and intriguing results featured in Sproncken's Ph.D. research demonstrate the potential of electrostatically co-assembled polymer-based soft matter systems for a wide variety of applications.
Materials
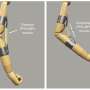
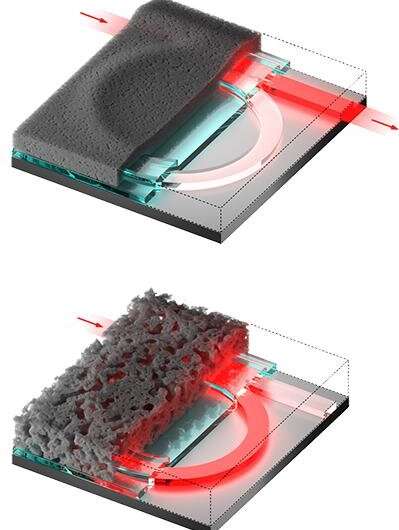
First, Sproncken prepared soft material coatings by dipping substrates in a number of solutions containing polyelectrolytes, which are polymers that have an electrolyte or charged group. The resulting polymer network can undergo swelling and collapsing, depending on the acidity (pH) of the solution that the network is exposed to.
"By manipulating this exposure process, it's possible to create a dry polymer film with very low or very high porosity," says Sproncken. "The refractive index of the material depends on the material porosity, which makes this material ideal for applications in photonics."
Indeed, Sproncken and Voets worked with Oded Raz and Mahir Mohammed from the department of Electrical Engineering at TU/e to create reprogrammable photonic microchips by coating microchips with the variable porosity material.
Ice off
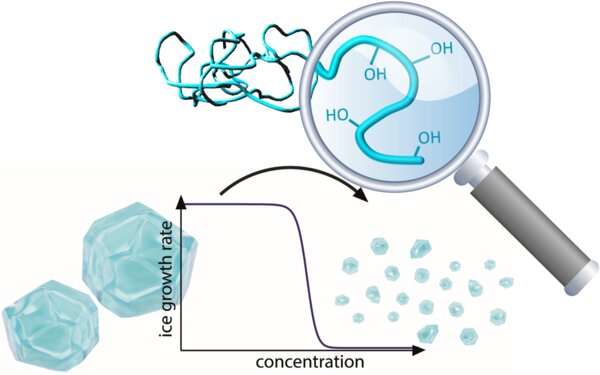
For the next polymer assembly method, Sproncken and his collaborators included an ice-binding polymer in micelles, or colloidal particles, with a polyelectrolyte complex core. Attaching uncharged polymers prevents the cores from growing or fusing into large aggregates of uncontrollable size.
When this specific polymer binds to ice crystals, it can slow the growth of the ice crystals. It paves the way for materials that could prevent undesirable ice growth on the windshields of cars or the wings of airplanes.
"We found that that the ice-binding activity of the polymer is maintained after inclusion in the micelles, making electrostatic co-assembly a promising tool as a basis for designing anti-icing materials," says Sproncken.
In the middle of a clock reaction
Finally, Sproncken explored ways to control the interaction between oppositely charged polymers in solution. Direct mixing of oppositely charged polymers often leads to phase separation where there is a polymer dense phase and a polymer dilute phase.
To stabilize the polyelectrolyte nanoscale structures, Sproncken used a reaction network to fix the size and shape of the polymeric nanoparticles. "The operation of this reaction network is twofold," notes Sproncken. "It tunes the interaction between the oppositely charged macromolecules while simultaneously stapling parts of the chains together."
In the future, such stable nanoparticles could be used as nanocarriers in biomedical applications, and it's all possible thanks to a so-called clock reaction that prevents large unusable aggregates from forming.
Future soft matter adventures
And as the clock winds down on Sproncken's time at TU/e, he's already preparing himself for his next soft matter adventure. He's set to move to the University of Fribourg in Switzerland where he'll work in the lab of Prof. Michael Mayer.
"I'm really looking forward to this next step in my research career, and to use the skills I have learned here at TU/e," he says. His postdoctoral research will focus on the assembly of block copolymer membranes for water two-phase systems.
More information: Thesis: Attractive Soft Matter: Polyelectrolyte Assembly for the Design of Functional Materials, research.tue.nl/en/publication … ly-for-the-design-of
Provided by Eindhoven University of Technology