Illuminating tissue formation through flourescence
Researchers at ETH Zurich have developed a molecule that fluoresces where new tissue is forming in the body. Alongside helping to detect tumors, the molecule could play a significant role in research of wound healing disorders.
Collagen is the most abundant protein in the human body. It makes up a third of protein content and single strands assemble to form stable fibers that give structure to connective tissue such as skin, tendons, cartilage and bones. Researchers at ETH Zurich have now developed a multi-component molecule that interacts with collagen and can be used to illuminate new tissue growth in the body.
Our bodies start producing more collagen as wounds heal—or as tumors grow. During this process, the fibrous collagen molecules cross-link to create stable fibers. This requires LOX enzymes, which oxidize certain sites in the collagen molecules. Subsequently, the chemically altered sites on different collagen strands react with each other, causing the strands to fuse together.
Sensor combined with functional peptide
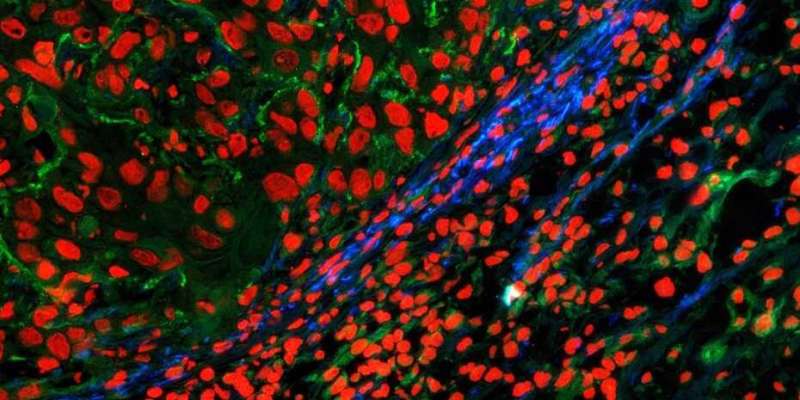
Led by Professor Helma Wennemers, a professor at the Laboratory of Organic Chemistry at ETH Zurich, the team of researchers developed a sensor molecule with inducible fluorescence. The molecule itself isn't fluorescent, but after reacting with the LOX enzyme, it begins to light up. In this way, the sensor molecule acts as a marker for LOX activity. Next, the scientists combined this molecule with a short fibrous peptide similar to collagen. They conjugated this peptide with a what is known as a reactive group that reacts with collagen only if the latter has been oxidized.
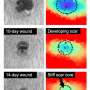
In collaboration with researchers from the group led by Sabine Werner, Professor of Cell Biology, the scientists conducted experiments with mice whose skin had been injected with the multi-component molecule. They also performed in vitro experiments with tissue sections. Their investigations revealed that the molecule anchors itself to collagen fibers where new tissue is being formed. And it lights up when new tissue starts growing and the LOX enzyme is being formed. "Thanks to its modular design with three components—the sensor, the peptide and the reactive group—our system is exceptionally specific and precise," says Matthew Aronoff, senior scientist in Wennemers' group and lead author of the study.
Applications in oncology and wound healing
Because new tissue forms primarily at the edges of tumors as they grow, one application for the new molecule is in biopsy examinations to show the boundaries of a tumor. "One of our visions is that surgeons will one day use this molecule in an operating theater when removing a tumor," Wennemers says. The molecule would show surgeons the boundary of the tumor and help them to remove it entirely.
Other potential applications for the new marker molecule are in the field of wound healing, for example to investigate tissue formation in general or healing disorders in patients suffering from diabetes or other diseases. Such questions are also addressed within the framework of the interdisciplinary skin research project Skintegrity, in which ETH Zurich is involved.
Having applied for a patent for their system, the scientists are currently exploring various options for bringing it to market and developing it for a wider range of applications.
More information: Matthew R. Aronoff et al, Imaging and targeting LOX-mediated tissue remodeling with a reactive collagen peptide, Nature Chemical Biology (2021). DOI: 10.1038/s41589-021-00830-6
Journal information: Nature Chemical Biology
Provided by ETH Zurich