Researchers reveal robust ethane-trapping porous organic cage for efficient ethylene purification application
The removal of ethane (C2H6) from its analogous ethylene (C2H4) is of great importance in the petrochemical industry, and is highly challenging due to their similar physicochemical properties. The use of emerging porous organic cage (POC) materials for C2H6/C2H4 separation is still in its infancy.
Calix[4]resorcinarene, a kind of macrocyclic cavitand with an intrinsic cavity and eigh polar upper-rim phenolic groups, has been documented as excellent building block to construct cage compounds with tunable cavities for encapsulating various guest molecules.
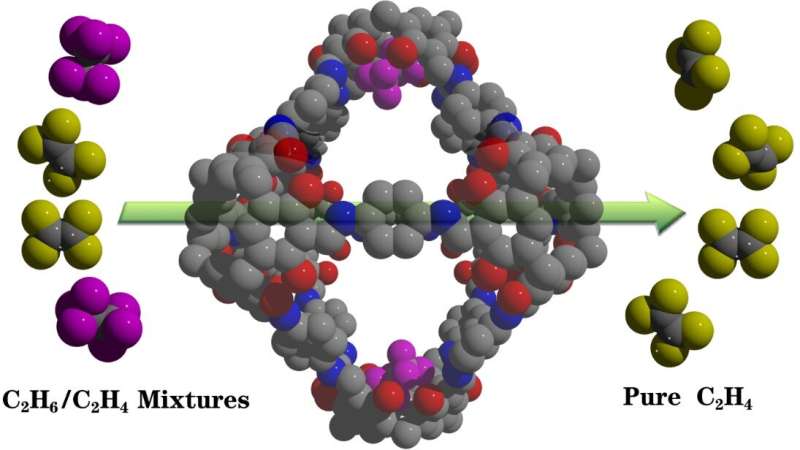
In a study published in Nature Communications, the research group led by Prof. Yuan Daqiang from Fujian Institute of Research on the Structure of Matter of the Chinese Academy of Sciences reported that a [6+12] octahedral calix[4]resorcinarene-based POC (CPOC-301) is an excellent C2H6 -selective material, and can be used as a robust absorbent to directly afford high-purity C2H4 from C2H6/C2H4 mixture.
The researchers prepared CPOC-301 via self-assembly of tetraformylresorcin[4]arene and p-phenylenediamine under mild condition.
Single-crystal X-ray diffraction revealed that CPOC-301 has a truncated octahedron structure, with eight trigonal ports having edge length reaching about 12 Å, and a large cavity with inner diameter as well as volume that respectively reach 16.8 Å and 4270 Å3. The solid-state packing of CPOC-301 suggested that it possesses a one-dimensional channel, with a diameter of ~7 Å, viewed from [001] direction.
The nitrogen (N2) gas adsorption isotherm of CPOC-301 showed a typical type I curve with a small fraction of a type IV adsorption behavior. The maximum N2 adsorption is 670 cm3 g-1, and the calculated Brunauer–Emmett–Teller (BET) of CPOC-301 is up to 1962 m2 g-1. Moreover, CPOC-301 exhibited preferential adsorption of C2H6 over C2H4 at room temperature.
The ideal adsorbed solution theory (IAST) calculation result demonstrated that C2H6/C2H4 selectivity range for CPOC-301 is from 1.3 to 1.4 at 293 K. The breakthrough curves further proved that CPOC-301 can efficiently realize the complete separation of C2H4 from C2H6/C2H4 mixtures, and its separation performance does not obviously change within seven continuous cycles.
Molecular modeling studies suggested the exceptional C2H6 selectivity is due to the suitable resorcin[4]arene cavities in CPOC-301, which form more multiple C–H···π hydrogen bonds with C2H6 than with C2H4 guests.
This study sheds light on the design and synthesis of POCs based on supramolecular cavitands as "porous additives" in column and membrane separation applications for industrially important gases in the future.
More information: Kongzhao Su et al, Efficient ethylene purification by a robust ethane-trapping porous organic cage, Nature Communications (2021). DOI: 10.1038/s41467-021-24042-7
Journal information: Nature Communications
Provided by Chinese Academy of Sciences