'Planar and curved' pyrrole-fused azacoronenes

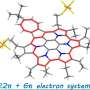
Recent study on synthetic approaches toward polycyclic aromatic hydrocarbons (PAHs) such as graphene with a well-defined structure have attracted much attention. A research group at Ehime University has been studying the synthesis and fundamental properties of pyrrole-fused azacoronene (HPHAC), a nitrogen-containing PAH. HPHACs are composed of electron-rich pyrroles, which are easily oxidized, and their dicationic species in particular exhibit unique features such as global aromaticity based on macrocyclic π-conjugation. However, all the compounds reported so far have bulky substituents at the periphery of the HPHAC skeleton, and thus the characterization of the physical properties of the pristine HPHAC itself or the π-electron function based on π-π interactions has not been possible.
In this study, the group has synthesized two new derivatives, one with alkyl groups at the periphery of the HPHAC skeleton and the other with concave π-planes above and below the HPHAC skeleton. The HPHAC with alkyl groups were found to be more readily oxidized than the compounds previously reported and to exhibit stable redox properties. In addition, reflecting its planar structure, the HPHAC forms an alternating stacked column structure with electron-deficient π-electron compounds. On the other hand, the HPHAC with an extended π-electron system exhibits an unusual π-electron function with a double concave surface. Reflecting its shape, this electron-rich HPHAC strongly interacts with spherical fullerene, which is an electron-deficient π-electron compound. A comparison of the global aromaticities of the two dicationic species reveals that the double-concave HPHAC possesses weaker aromaticity.
Recent research on π-electron materials has led to the synthesis of three-dimensional structures, such as bowl- and saddle-shaped compounds, instead of the conventional planar ones. However, there have been few studies focusing on the π-electron function of the 3-D structure, except for clarifying its structural features using single crystal X-ray structure analysis. By elucidating the detailed structure-property relationships between analogues that share the same skeleton but have different 3-D structures, new design guidelines can be obtained for organic electronics and spintronics materials involving π-electron functions.

More information: Yoshiki Sasaki et al, Radially π-Extended Pyrrole-Fused Azacoronene: A Series of Crystal Structures of HPHAC with Various Oxidation States, The Journal of Organic Chemistry (2021). DOI: 10.1021/acs.joc.0c02825
Provided by Ehime University