Balancing between build-up and break-down of bone
Despite what some people think, bone is not merely a passive component of the body. The skeleton is structurally dynamic and responds to life's physical stresses with continual equilibration between bone mass loss and reformation. This ensures healing and remodeling in tune with the ebb and flow of calcium and phosphorus in the bloodstream. Now, researchers at Osaka university have identified a molecule—secretory leukocyte protease inhibitor (SLPI)—that helps mediate this critical balance, which could be used in the development of new treatments for bone diseases such as osteoporosis.
Skeletal tissue changes are orchestrated primarily by parathyroid hormone (PTH), a regulator of blood calcium levels that is secreted by the parathyroid glands in the neck. PTH is known to have a dual effect on bone—its action is primarily catabolic, causing bone dissolution and removal. However, in small intermittent doses, PTH can also increase bone mass (anabolic). Though PTH has long been used for the clinical treatment of osteoporosis, the precise mechanism and pathways whereby PTH promotes bone formation are poorly understood.
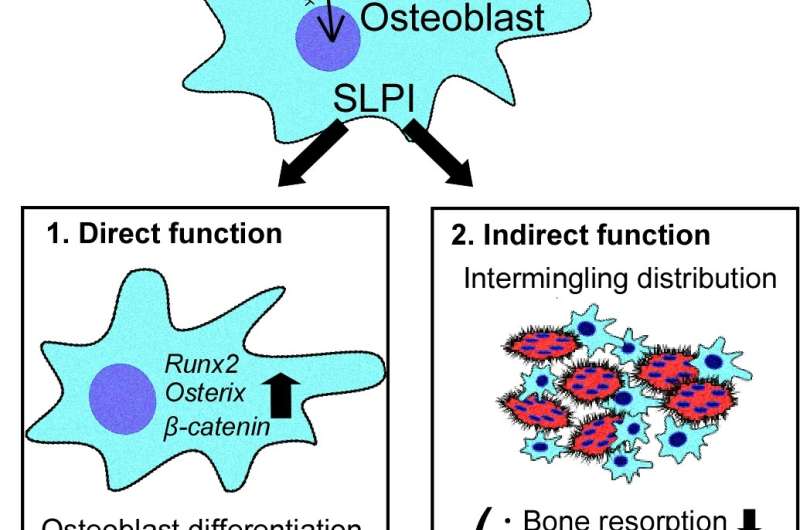
The researchers looked at the interactions between cells that mediate bone formation (osteoblasts), cells that mediate bone loss (osteoclasts) and the functional role of SLPI in bone metabolism in vivo. Akito Morimoto, lead author, explains the research methodology of the new study published in Nature Communications: "We could establish that PTH highly upregulates the gene Slpi in osteoblasts in animal models. We analyzed the bone phenotype of experimental mice in which the gene was knocked out and showed that genetic modification of Slpi prevented PTH from inducing bone formation. Moreover, Slpi induction in osteoblasts themselves increased their differentiation while promoting osteoblast-osteoclast contact which reduces bone loss activity." Furthermore, biomicroscopic imaging in living bone demonstrated that SLPI secreted outside the cells is essential for association between osteoblasts and osteoclasts and the cell-cell interactions that PTH mediates.
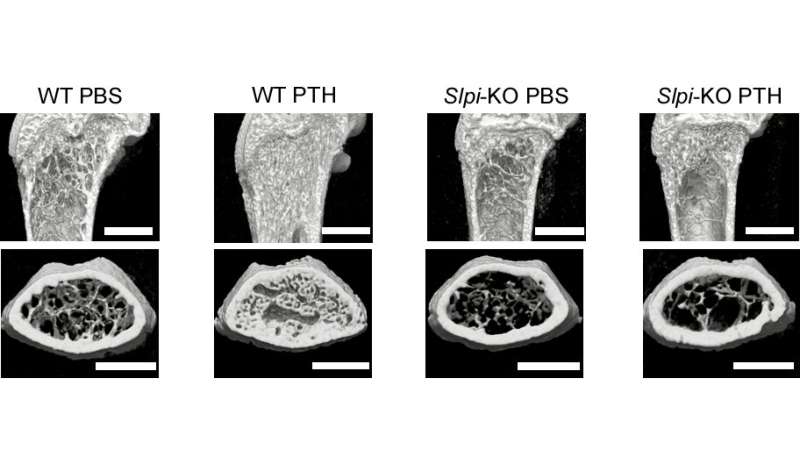
Corresponding author Junichi Kikuta summarizes their results. "Our findings clarify the roles of SLPI as a novel coupling factor and coordinator of bone remodeling for conservation of mass, strength and structural integrity. Not only does it promote bone formation by osteoblasts, it also attracts osteoclasts closer to osteoblasts to suppress bone loss."
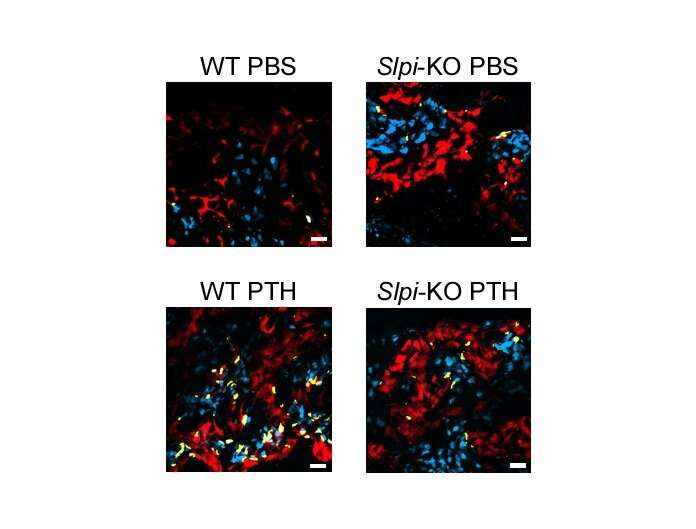
"A clear understanding of the cellular networks and molecular pathways that mediate PTH anabolism will enhance clinical applicability of this drug," senior author Masaru Ishii explains. "Moreover, it may inform the development of innovative pharmacotherapies for managing osteoporosis and other intractable orthopedic diseases."
The article, "SLPI is a critical mediator that controls PTH-induced bone formation" was published in Nature Communications.
More information: "SLPI is a critical mediator that controls PTH-induced bone formation" Nature Communications (2021). DOI: 10.1038/s41467-021-22402-x
Journal information: Nature Communications
Provided by Osaka University