New insights put a freeze on the mechanisms for safely cryopreserving biological materials
Cryopreservation involves preserving biological materials, such as cells, tissues, and organs, at ultra-low temperatures so that they can be revived and used at a later date. To achieve cryopreservation such that the preserved materials are not damaged, scientists use various chemicals called cryoprotectants, which facilitate the freezing process. Unfortunately, many of the existing cryoprotective agents have major limitations. For example, dimethyl sulfoxide is useful for cryopreserving red blood cells, but it is also toxic to the cells.
To overcome these limitations, researchers at the Japan Advanced Institute of Science and Technology (JAIST) and Japan's RIKEN have experimented with a class of chemicals known as polyampholytes, identifying one polyampholyte named carboxylated ε-poly-ʟ-lysine (COOH-PLL) as a promising cryoprotectant that does not have toxic effects on cells.
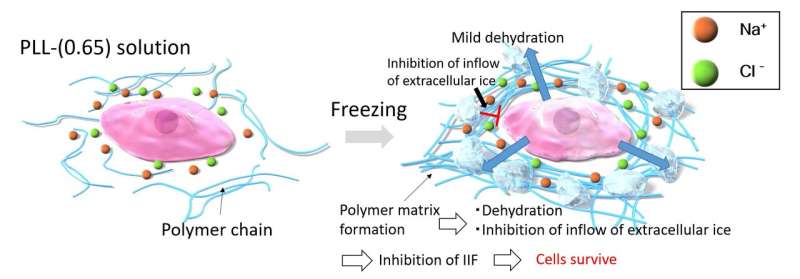
However, the mechanism by which COOH-PLL acts as a cryoprotectant remained poorly understood, and this hindered efforts at developing better polyampholyte cryoprotectants. To address this knowledge gap, this team of researchers, which was led by Professor Kazuaki Matsumura from JAIST—and which also included Asst. Prof. Robin Rajan from JAIST and Dr. Fumiaki Hayashi and Dr. Toshio Nagashima from RIKEN—conducted another study in which they used a method called solid-state nuclear magnetic resonance (NMR) spectroscopy to characterize what happens to water molecules, sodium and chloride ions, and a form of COOH-PLL called PLL-(0.65) when they are mixed and frozen to temperatures used in cryopreservation. Their findings appear in a recent issue of the peer-reviewed journal Communications Materials.
As expected, the NMR signals recorded from these frozen solutions indicated reduced molecular-level mobility and increased solution viscosity (i.e., increased resistance to flow) at ultra-low temperatures. Importantly, the PLL-(0.65) molecules trapped the water molecules and ions in ways that prevented the formation of intracellular ice crystals and countered the effects of osmotic shock (a form of physiological dysfunction in which a sudden change in ion concentrations around a cell can lead to rapid inflow or outflow of fluid into or from the cell, damaging it). Intracellular ice crystals and osmotic shock are major causes of cellular or tissue damage during freezing, so the ability of PLL-(0.65) to prevent both helps to explain its effectiveness as a cryoprotectant.
In describing the scientific value of these findings, Prof. Matsumura notes that the use of NMR "enabled the characterization of the cryoprotective properties of polymers that operate through mechanisms different from those of current cryoprotectants." He predicts that this characterization "will facilitate the molecular design of new cryoprotectants," which will "promote the clinical use of polymeric cryoprotective agents that can serve as efficient alternatives to small molecule agents such as dimethyl sulfoxide and glycerol."
Small molecule cryoprotectants are only useful for preserving cells and small tissues, but polymeric cryoprotectants may enable researchers to achieve the cryopreservation of larger tissue volumes or even whole organs. This would be an important advance for emerging fields such as regenerative medicine, which aims to restore normal physiological functions by replacing or regenerating cells, tissues, and organs.
The team's findings may also have important implications for basic biology. In recent years, scientists have learned that intrinsically disordered proteins, which lack a fixed three-dimensional shape, serve an important role in protecting cells from damage related to desiccation and osmotic shock at low temperatures. Because proteins are themselves a class of polyampholytes, these recently published findings concerning COOH-PLL's behavior at ultra-low temperatures may help to explain the mechanisms by which intrinsically disordered proteins protect cells. Further research into the mechanisms of such proteins may in turn lead to the development of advanced materials with uses in regenerative medicine.
In conclusion, these findings may be of considerable value to biology and medicine. "Based on our now-improved understanding of mechanisms of polymeric cryoprotective agents," notes Prof. Matsumura, "we expect to develop new cryopreservation technologies for large tissue volumes and organs, which will permit the industrial-scale implementation of regenerative medicine via the establishment of banks of engineered tissues." These steps towards regenerative medicine will do much to improve the lives of people living with medical disorders that are currently incurable.
More information: Molecular mechanisms of cell cryopreservation with polyampholytes studied by solid-state NMR. Communications Materials. DOI: 10.1038/s43246-021-00118-1
Provided by Japan Advanced Institute of Science and Technology